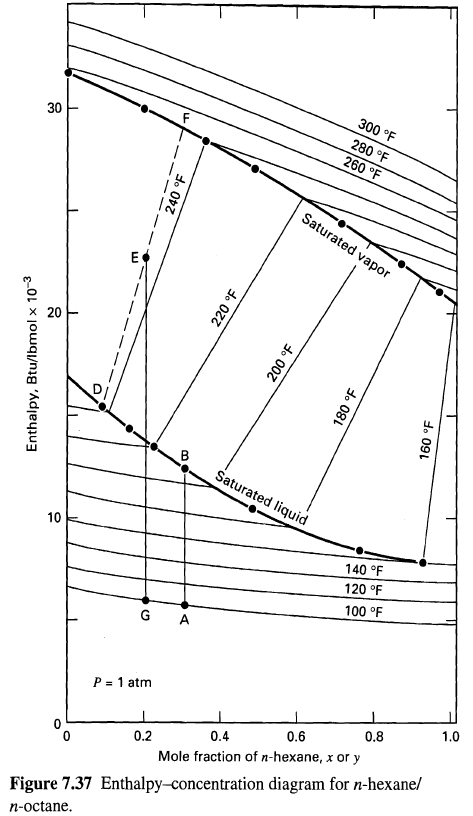
An enthalpy-concentration diagram is given in Figure for a mixture of n-hcxanc (H). and n-octane (0) at
Question:
An enthalpy-concentration diagram is given in Figure for a mixture of n-hcxanc (H). and n-octane (0) at 101 kPa. Using this diagram, determine the following:
(a) The mole-fraction composition of the vapor when a liquid containing 30 mol% H is heated from point A to the bubble-point temperature at point B.
(b) The energy required to vaporize 60 mol% of a mixture initially at l00oF and containing 20 mol% H (point G).
(c) The compositions of the equilibrium vapor and liquid resulting from part(b).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





