An ideal gas (1.0 mol) is the working substance in an engine that operates on the cycle
Question:
An ideal gas (1.0 mol) is the working substance in an engine that operates on the cycle shown in Figure; processes BC and DA are reversible and adiabatic.
(a) Is the gas monatomic, diatomic, or polyatomic?
(b) What is the engine efficiency?
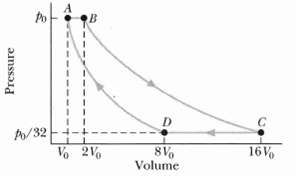
Transcribed Image Text:
A/32 -4. Vo 2V 8V Volume 16V Pressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a Using Eq 1954 for process D A gives PDV PAVA Po 8V Pov 32 which leads to 8 32 y53 The result see ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A four-cylinder two-stroke 2.4-L diesel engine that operates on an ideal Diesel cycle has a compression ratio of 22 and a cutoff ratio of 1.8. Air is at 70oC and 97 kPa at the beginning of the...
-
A four-cylinder, two-stroke 2.4-L diesel engine that operates on an ideal Diesel cycle has a compression ratio of 22 and a cutoff ratio of 1.8. Air is at 70C and 97 kPa at the beginning of the...
-
A four-cylinder, four-stroke, 4.5 L diesel engine that operates on an ideal diesel cycle has a compression ratio of 17 and a cutoff ratio of 2.2. Air is at 27 and 97 kPa at the beginning of the...
-
The per-unit cost of an item is its average total cost (5 total cost/quantity). Suppose that a new cell phone application costs $100,000 to develop and only $.50 per unit to deliver to each cell...
-
What is meant when we say that the trade theory discussed in previous chapters is static in nature? What is meant by comparative statics?
-
Go to the Web site of Airbus A38G, the biggest passenger plane ever built (525 passengers or more on 2 floors, with a range of 15,000 km, and a wingspan of 80 metres), www.airbus....
-
Fill in the Blank. In the finite element method, the elements are assumed to be interconnected at certain points known as ____________ .
-
Koch Construction Company began operations on January 1, 2015, when it acquired $15,000 cash from the issuance of common stock. During the year, Koch purchased $3,000 of direct raw materials and used...
-
Lopez Company is considering replacing one of its old manufacturing machines. The old machine has a book value of $ 4 7 , 0 0 0 and a remaining useful life of four years. It can be sold now for $ 5 7...
-
Based on past sales experience, an appliance store stocks five window air conditioner units for the coming week. No orders for additional air conditioners will be made until next week. The weekly...
-
Figure shows a reversible cycle through which 1.00 mol of a monatomic ideal gas is taken. Assume that p = 2p0, V = 2V0, p0 = 1.01 x 105 Pa, and V0 = 0.0225 m3. Calculate (a) The work done during the...
-
The efficiency of a particular car engine is 25% when the engine does 8.2 kJ of work per cycle. Assume the process is reversible. What are? (a) The energy the engine gains per cycle as heat Qgain...
-
We are often interested in modeling the growth and spread of a population of bacteria. While we often have a good idea of bacterial reproduction rates, getting a handle on how fast they spread is...
-
You deposit $3000 in an account earning 4% interest compounded monthly. How much will you have in the account in 15 years? $
-
Suppose you want to have $600,000 for retirement in 25 years. Your account earns 5% interest. How much would you need to deposit in the account each month? Submit Question
-
What is the difference between HTML and XML, how can attackers make use of XML to attack a server?
-
You have $300,000 saved for retirement. Your account earns 9% interest. How much will you be able to pull out each month, if you want to be able to take withdrawals for 15 years? $ Submit Question
-
You can afford a $400 per month car payment. You've found a 5 year loan at 7% interest. How big of a loan can you afford? $ Submit Question
-
A particle carrying \(3.00 \mathrm{nC}\) of positive charge is at the origin of a rectangular coordinate system, and a particle carrying \(3.00 \mathrm{nC}\) of negative charge is on the \(x\) axis...
-
A heat engine has a heat input of 3 Ã 104 Btu/h and a thermal efficiency of 40 percent. Calculate the power it will produce, in hp. Source 3 x 10 Btu/h 40% HE Sink
-
Starting with the definitions of momentum and kinetic energy, derive an equation for the kinetic energy of a particle expressed as a function of its momentum.
-
A metal tank with volume 3.10 L will burst if the absolute pressure of the gas it contains exceeds 100 atm. (a) If 11.0 mol of an ideal gas is put into the tank at a temperature of 23.0oC, to what...
-
Three moles of an ideal gas are in a rigid cubical box with sides of length 0.200 m. (a) What is the force that the gas exerts on each of the six sides of the box when the gas temperature is 20.0oC?...
-
With the assumptions of Example 18.4 (Section 18.1), at what altitude above sea level is air pressure 90% of the pressure at sea level?
-
Solve these equations Sy = b with 81, 82, 83 in the columns of S: 0 0 10 Y2 100 0 1998-8-1988-8 and S is a sum matrix. The sum of the first 5 odd numbers is ][
-
Active Life Ltd has decided to manufacture a new line of running shoes based on a $50,000, two-year feasibility study. The shoes will sell for $85 a pair and has a variable cost of $30 a pair. It is...
-
Arch Airway expects to purchase 5.6 million gallons of jet fuel in three months and decides to use heating oil futures to hedge the fuel price risk. A regression has been performed: The dependent...

Study smarter with the SolutionInn App


