Arrange these alkenes in order of increasing rate of reactio0n with HCI: CH, CH2=CH2 a) CH;CH2CH=CH2 CH;CH,C=CH,
Question:
Arrange these alkenes in order of increasing rate of reactio0n with HCI:
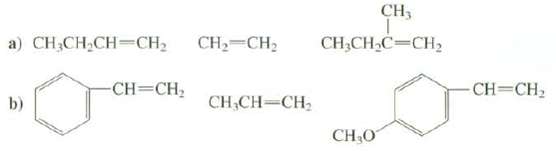
Transcribed Image Text:
CH, CH2=CH2 a) CH;CH2CH=CH2 CH;CH,C=CH, -CH=CH; CH=CH2 CH,CH-CH b) CHO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a CHCH slowest CH...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Arrange the following alkenes in order of increasing stability:
-
Arrange the following compounds in order of increasing boiling point. Explain your answer in terms of the intermolecular forces in each compound. (a) (b) (c) (d)
-
Arrange the following compounds in order of increasing boiling point. (b) (a) (d) (c)
-
Maynard Appliances is holding a Fifty- Fifty Sale. Major appliances may be purchased for nothing down and no interest to pay if the customer pays 50% of the purchase price in six months and the...
-
Faced with rising pressure for a $15 per hour minimum wage rate, the farming industry is currently exploring the possible use of robotics to replace some farm workers. The Lettuce Bot is one such...
-
A honeybee with a mass of 0.150 g lands on one end of a floating 4.75-g popsicle stick, as shown in FIGURE 9-42. After sitting at rest for a moment, it runs toward the other end with a velocity...
-
Suppose $U(x)$ is a utility function with Arrow-Pratt risk aversion coefficient $a(x)$. Let $V(x)=c+b U(x)$. What is the risk aversion coefficient of $V$ ?
-
Patricia Fox plans to borrow $5,000 and to repay it in 36 monthly installments. This loan is being made at an annual add-on interest rate of 7.5 percent. a. Calculate the finance charge on this loan,...
-
Pore Networks in Membranes. (20 pts) Consider two membranes below, with equal void space, but differently configured. The membrane on the left consists of a narrow and a wide pore in parallel, while...
-
Fruity Juices, Inc. produces five different flavors of fruit juice: apple, cherry, pomegranate, orange, and pineapple. Each batch of product requires processing in three departments (blending,...
-
Consider a multidatabase system in which every local site ensures local serializability, and all global transactions are read only. a. Show by example that nonserializable executions may result in...
-
Show the structure of the carbocations that are formed in the reaction of HBr with 2-hexena and explain why two products are formed.
-
Find the composite function g(h(x)), where g(u) 1 2u + 1 and h(x) = x + 2 2x + 1
-
Question 2 A consumer purchases two goods, food (x) and clothing (y).He has the utility function U(X,Y) = XY, where X and Y denote amounts of X and Y consumed. Marginal utilities of X and Y are MU x...
-
1. As a financial analyst, you plan to help Arkansas Tech Endowment choose a fund. The risk-free rate of return is 4%. The table below shows a basic statistic summary for two potential funds: Odyssey...
-
Explain the rule for determining the optimal resource use for a single resource or input and determine the optimal amount of labor, given that each output sells at $2 and labor is $26 per unit. Labor...
-
Consider a small open economy (e.g. the Netherlands) producing two goods, clothing and food. The clothing industry uses capital (K) and labor (LC) as inputs, while the food industry uses land (La)...
-
Taxation Systems for Business Entities Why do we tax business entities instead of just taxing their owners? What is the difference between the 'corporate taxation' model and the 'flow-through' model...
-
In March 1963, Ironman was first introduced in issue number 39 of Tales of Suspense. The original price for that issue was 12 cents. By March 2012, 49 years later, the value of this comic book had...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Write equations for the stepwise formation of each of the following complex ions. a. Ni(CN) 4 2- b. V(C2O 4 ) 3 3 -
-
The benzyl ammonium ion (C6H5CH2NH3) has pKa 9.33, and the propyl ammonium ion has pKa = 10.71. Which is the stronger base, benzyl amine or propylamine? What are the pKbS of benzyl amine and...
-
Without looking at Table 24.2, rank the following compounds in order of ascending basicity. (a) p-Nitro aniline, p-aminobenzaldehyde, p-Bromoaniline (b) p-Chloroanilinc, p-aminoacetophenone, p-methyl...
-
Calculate the percentages of neutral and protonated forms present in a solution of 0.0010 M pyrimidine at pH = 7.3. The pKa of pyrimidinium ion is 1.3.
-
Chang Company provided the following data for this year: Manufacturing overhead costs incurred: Indirect materials Indirect labor Property taxes, factory Utilities, factory Depreciation, factory...
-
ces On January 1, 2024, the Mason Manufacturing Company began construction of a building to be used as its office headquarters. The building was completed on September 30, 2025. Expenditures on the...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...

Study smarter with the SolutionInn App


