Assign R or S configuration to the following molecule, write the product you would expect from SN2
Question:
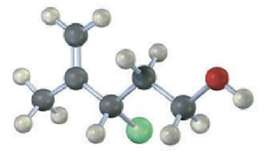
Assign R or S configuration to the following molecule, write the product you would expect from SN2 reaction with NaCN, and assign R or S configuration to the product (yellow-green =Cl):
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
CH HH CHOH NaCN H3C CH HH R NC H CH2OH NaCl HCI T...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to the chirality center in the following molecular model of the amino acid methionine (blue = N, yellow =S):
-
Assign R or S configuration to the chirality center in each of the followingmolecules: CH (c) . (b) (a) 2 "- HS efpo -- CH-
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
In the Walton Bookstore example with a discrete demand distribution, explain why an order quantity other than one of the possible demands cannot maximize the expected profit.
-
Evaluate Brian Conrad's approach to appraisal interviews. Write a paragraph or two summarizing what Conrad is doing well and how he might further improve the effort.
-
Add any unshared electrons to give each element an octet in its valence shell in the formulas below and indicate any formal charges. All of the hydrogen atoms that are attached to heteroatoms have...
-
Which of the following would prevent a paid disbursement from being paid a second time? a. Individuals responsible for signing checks should prepare vouchers. b. Disbursements should be approved by...
-
The partially completed inventory record for the table top subassembly in Figure shows gross requirements scheduled receipts, lead time, and current on-hand inventory. a. Complete the last three rows...
-
You have a ball bearing and a bowl.You let the ball roll down from the top of the ball; it moves up and down the bowl's wall for some time. (a) Is this vibrational motion? explain why or why not. (b)...
-
The following table contains data for a hypothetical economy: Consumer Expenditures $500 million Wages $650 million Investment Expenditures $200 million Interest Payments $100 million Government...
-
From what alkyl bromide was the following alkyl acetate made by SN2 reaction? Write the reaction, showing allstereochemistry.
-
Draw the structure and assign Z or F stereochemistry to the product you expect from E2 reaction of the following molecule with NaOH (yellow-green =Cl):
-
Assume that you are testing the alternative hypothesis H a : m > 54.2. If the sample mean has a standard score of z = -1.0, what do you conclude? Why is it not necessary to actually conduct a formal...
-
What are some differences between physical library and virtual legal resources ? When might it be better to visit a physical library compared to a virtual legal resources and vice versa? In what type...
-
Under which rule(s) of the supreme court civil rules, B.C. Reg. 168/2009, a party to a proceeding can make an offer to settle? Make sure you list the appropriate section(s) and explain the...
-
Explain what is meant by the term gross negligence (IN YOUR OWN WORDS) and provide four examples of situations in sports in which the conduct of a defendant might rise to this level of negligence....
-
Dr. Daniela Yeung, a health psychologist, has been conducting a federally funded ethnographic study of couples in which the male partner has been imprisoned for intimate partner violence. Over the...
-
1. A cart with a mass of 25.0 kg is rolling with a speed of 14 m/s. What is the magnitude of the cart's momentum? 2. Using the concept of impulse, explain why an egg wont break when thrown into a...
-
Peggy, standing on a moving railroad car, passes Ryan at velocity v. Peggy and Ryan both measure the length of the car, from one end to the other. The length Peggy measures is ___ the length Ryan...
-
Rosalie owns 50% of the outstanding stock of Salmon Corporation. In a qualifying stock redemption, Salmon distributes $80,000 to Rosalie in exchange for one-half of her shares, which have a basis of...
-
Consider the reaction: If a solution initially contains 0.210 M HC 2 H 3 O 2 , what is the equilibrium concentration of H 3 O + at 25 C? HCH3O (aq) + HO(1) H3O+(aq) + CH3O (aq) K 1.8 x 10-5 at 25 C =
-
Draw all four resonance forms of the fragment at m/z 73 in the mass spectrum of pentanoic acid.
-
Name the following carboxylic acid derivatives, giving both a common name and an IUPAC name where possible. (a) PhCOOCH2CH(CH3)2 (b) PhOCHO (c) PhCH(CH3) COOCH3 (d) PhNHCOCH2CH(CH3)2 (e) CH3CONHCH2Ph...
-
(a) Show how you would use acetic anhydride and an appropriate alcohol or amine to synthesize (i) benzyl acetate, (ii) N,N-diethylacetamide. (b) Propose a mechanism for each synthesis in part (a).
-
write a page on global migration giving statistics and then focus on migration within Africa and then South Africa. Write reference?
-
Lesotho'footwear industry manufacturer one in maseru and other in maputsoe.aputsoe.in maseru te marginal benefit associated wih ppolluiio cleeanup is MB = 3 0 0 - 1 0 Q while in maputsoe te marginal...
-
Under what conditions would the Fed choose to decrease the money supply, how would it do so , and what is the goal of doing so ? How does the Fed factor inflation into its actions?

Study smarter with the SolutionInn App


