Assign R, S configuration to each chirality center in the following molecular model of the amino acid
Question:
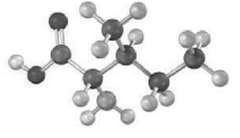
Assign R, S configuration to each chirality center in the following molecular model of the amino acid isoleucine (blue =N):
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (17 reviews)
HOC H3C ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to each chirality center in the following monosaccharide?s and tell whether each is a D sugar or an L sugar: (b) (a) - (c) - - - - - - - - - - CH- CH2 CH2
-
Assign R or S configuration to each chirality center in the followingmolecules: (b) NH2 (a) . "C r "O
-
Assign R or S configuration to each chirality center in prostaglandin E2 (Figure) the most abundant and biologically potent of mammalianprostaglandin RS .CO2H Arachidonic acid PGH2 H. RS RS " " ...
-
What requirements must be met to deduct a trade or business expense? an expense related to the production of income?
-
Show that the ideal efficiency is 90% for an engine in which fuel is heated to 3000 K and the surrounding air is 300 K? Ideal efficiency = Tbot - Tcold / Thot
-
Consider the two beams below; they are loaded the same but have different support conditions. Which beam has the larger maximum moment? First, find support reactions, then plot axial force (N), shear...
-
What four categories of KPIs are typically used to measure the success of an EA? Give two examples of each.
-
Last year, a toy manufacturer introduced a new toy truck that was a huge success. The company invested $2.5 million for a plastic injection molding machine (which can be sold for $2.0 million) and...
-
A shaft simply supported by a truncated conical pivot bearing that carries an axial load at its center and the cone angle is 120. The axial load on the bearings is 3kN. The shaft speed is 1440 rpm...
-
On January 1, 2014, Prince Corporation acquired 70% of the 100,000 outstanding voting shares of Song Limited for a cash consideration of $1,015,000. On that date, shares of Song Limited were trading...
-
Chloramphenicol, a powerful antibiotic isolated in 1949 from the Streptomyces Venezuelae bacterium, is active against a broad spectrum of bacterial infections and is centers in Chloramphenicol. ...
-
Which of the following structures represent mesocompounds? C (b) . (d) (c) (a) Br CH . Br .
-
Raoul Pictet, the Swiss physicist who first liquefied oxygen, attempted to liquefy hydrogen. He heated potassium formate, KCHO 2 , with KOH in a closed 2.50-Lvessel. KCHO 2 (s) + KOH(s) K2CO 3 (s) +...
-
Abed says he has written a system of two linear equations that has an infinite number of solutions. One of the equations of the system is y=3x-1. What could be the other equation?
-
jackie is standing 250 feet away from a large redwood tree. she looks up at the top of the tree and measures the angle of elevation to be 52. how tall is the tree
-
A corner store sells 60-watt bulbs in packs of 4. Write an equation that shows how the number of bulbs in stock, y, depends on the number of packages, x.
-
A gardener can plant 17 seeds in 25 minutes. At that rate, how long will it take him to plant 51 seeds?
-
A ski slope has an angle of elevation of 15.1 and a vertical drop of 1800 feet. Estimate the length of the ski slope?
-
The G string on a guitar is 59 cm long and has a fundamental frequency of 196 Hz. A guitarist can play different notes by pushing the string against various frets, which changes the strings length....
-
Assume that a trial balance is prepared with an account balance of $21,360 listed as $21,630 and an account balance of $1,500 listed as $15,000. Identify the transposition and the slide.
-
A gas sample at STP contains 1.15 g oxygen and 1.55 g nitrogen. What is the volume of the gas sample? a) 1.26 L b) 2.04 L c) 4.08 L d) 61.0 L
-
Predict the product(s) in each of the following aldol condensations. acetophenone + hexanal + hexanal NH
-
Some of the following molecules can be synthesized in good yield using an aldol condensation. Identify these and give the structures of the required starting materials. Others cannot be synthesized...
-
Write the structure that corresponds to each of the following abbreviations. (a) i-Pr-Ph (b) Pr-OH (c) Ac-Ph
-
What role do social institutions play in shaping individual and collective identities, and how do these identities intersect with race, gender, sexuality, and other aspects of social diversity to...
-
Blossom Ltd., which follows ASPE had the following comparative statement of financial position: Blossom Ltd. Comparative Statement of Financial Position December 31 Assets 2024 2023 Cash $84.000...
-
16. Let $V=\operatorname [span]\left\{\left(\begin{array}{1}1 \\ 0 \\ 1 \\ 0\end{array} ight), \left(\begin{array} (1)1 \\ 1 \\ 0 \\ 1\end{array} ight), \left(\begin{array} {1}0 \\ 1 \\ 1 AV...

Study smarter with the SolutionInn App


