Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as
Question:
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as shown in figure. At what positions in mevalonate would the isotopic labelappear?
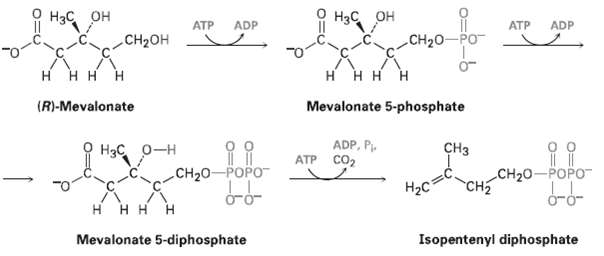
Transcribed Image Text:
Нзс, он H3C OH ATP ADP ATP ADP CH20-PO CH2OH н ннн H H H H Mevalonate 5-phosphate (R)-Mevalonate ADP, P. ATP Co2 CHз Hас о-н CH20-POPO CH2 CH20-POPO- H2C H H H H Mevalonate 5-diphosphate Isopentenyl diphosphate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
COAS CH3 COAS CH3 H3COH ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assume that acetyl COA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in lanosterol where...
-
For each Fischer projection, label each asymmetric carbon atom as (R) or (S). (a)-(f) the structures in Problem 5-18 In problem 5-18 (a to f) (a) (b) (c) (d) (e) (f) (g) (h) (i) CH,OH CH,OH Br CH,OH...
-
Starting with acetyl chloride, what nucleophile would you use to make each of the following compounds? a. b. c. d. e. f. CH COCH2CH2CH3 CH,CNHCH2CH3 CHCN(CH3)2 CH,COH NO2 CH,CO-
-
1. What is your brand/product? 2. Who is your target segment? 3. What are their needs/wants? 4. What message do you want to deliver to them?
-
1. What makes a CEO's job so complex? Use the mini-case to provide examples that help support your answer. 2. Is it a good practice to rehire a former CEO who has retired? Please explain the...
-
Given the information provided in Fig. 4.116, determine: In Figure 4.116 (a) IC. (b) VE. (c) VB. (d) R1. 0 18 V 4.7 k R, I'e 5.6 k 1.2 k
-
The specifications for a certain mass-produced valve prescribe a testing procedure according to which each valve can be classified as satisfactory or unsatisfactory (defective). Past experience has...
-
Beckett Industries manufactures a popular interactive stuffed animal for children that requires two computer chips inside each toy. The company pays $ 3 for each computer chip. To help to guard...
-
A cannon is fired with muzzle velocity of 158 m/s at an angle of elevation = 46. Gravity = 9.8 m/s 2 . a) What is the maximum height the projectile reaches? b) What is the total time aloft? c) How...
-
The balance sheets of Forest Company and Garden Company are presented below as at December 31, Year 8. Additional Information Forest acquired 90% of Garden for $207,900 on July 1, Year 1, and...
-
Assume that the three terpenoids in Problem 27.24 arc derived biosynthetically from isopentenyl diphosphate and dimethyl allyl diphosphate, each of which was isotopically labeled at the...
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in ?-cadinol where...
-
Draw Fischer projections of the following molecules: (a) The S enantiomer of 2-bromobutane (b) The R enantiomer of alanine, CH3CH (NH2) COOH (c) The R enantiomer of 2-hydroxypropanoic acid (d) The S...
-
Consider a greedy strategy for the following problem: We have a company with n workers. Worker w; works a shift (si, fi), where s; is that worker's start time and fi the finish time. We want to form...
-
Sheffield Book Warehouse Ltd. distributes hardcover books to retail stores. At the end of May, Sheffield's inventory consists of 230 books purchased at $17 each. Sheffield uses a perpetual inventory...
-
Calculate the envelope set (frontier) for the following four assets giving a constant of 0.00 to determine portfolio x's optimal weights, and a constant of 0.04 to determine portfolio y's optimal...
-
Alec, Daniel, William, and Stephen decide today to save for retirement. Each person wants to retire by age 60 and puts $10,600 into an account earning 9% compounded annually. Note: Use tables, Excel,...
-
Answer the following questions. Define and diagram the value chain. Is there such a thing as the first-mover advantage? Why or why not? What are Porter's "five forces"?
-
Aprinted circuit board is produced by passing through a sequence of three steps. The scrap rates for steps one through three are 5 percent, 3 percent, and 3 percent, respectively. If 10,000 good...
-
Subtract the polynomials. (-x+x-5) - (x-x + 5)
-
Complete the table. (All solutions are at 25 C.) [H3O+] 3.7 X 10- [OH-] 1.6 X 10-11 pH 3.15 11.1 Acidic or Basic
-
Using a three-monomer segment, write an equation for a. The reaction of polystyrene with Cl2 + FeCl3 b. The reaction of polystyrene with HNO3 + H2SO4
-
Methyl methacrylate (Table 14.1) can be polymerized by catalytic amounts of n butyllithium at 78C. Using eqs. 14.17 and 14.18 as a model, write a mechanism for the reaction. Show how the intermediate...
-
Ethylene oxide can be polymerized by base to give carbowax, a water-soluble wax. Suggest a mechanism for the reaction. CH,-CH, HOCH OCH2CH2OH ethylene oxide
-
Suppose the correlation between the stock euro returns of Siemens and the USD/EUR exchange rate is 0.2. The standard deviation of the USD/EUR is 10% and the standard deviation of Siemens's stock euro...
-
list and describe the three key client-related factors that the advisor is required to consider when developing a "suitable" investment portfolio for their client. Please cite resources used
-
Year 1 2 3 Amount ($) 2000 3000 4000 An investment made today will pays you the above cash flows at the end of each year. If your required rate of return is 5% annual interest, how much will you pay...

Study smarter with the SolutionInn App


