At point D in Figure the pressure and temperature of 2 mol of an ideal monatomic gas
Question:
At point D in Figure the pressure and temperature of 2 mol of an ideal monatomic gas are 2 atm and 360 K. The volume of the gas at point B on the PV diagram is three times that at point D and its pressure is twice that at point C. Paths AB and CD represent isothermal processes. The gas is carried through a complete cycle along the path DABCD. Determine the total amount of work done by the gas and the heat supplied to the gas along each portion of thecycle.
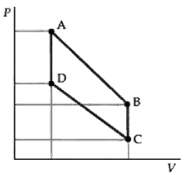
Transcribed Image Text:
B.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The volume at D from V nRT P V D 2 8314 360202 L 296 L We f...View the full answer

Answered By

Muhammad Salman Alvi
Well, I am a student of Electrical Engineeing from Information Technology University of Punjab. Just getting into my final year. I have always been good at doing Mathematics, Physics, hardware and technical subjects. Teaching profession requires a alot of responsibilities and challenges.
My teaching experience started as an home tutor a year ago. When I started teaching mathematics and physic subjects to an O Level student. He was about 14 years old. His name was Ibrahim and I used to teach him for about 2 hours daily. Teaching him required a lot of patience but I had to be polite with him. I used to give him a 5 min break after 1 hour session. He was quite weak in basic maths and calculation. He used to do quite a lot of mistakes in his homework which I gave him weekly. So I decided to teach him basics from scratch. He used to say that he got the concept even if he didn't. So I had to ask him again and again. I worked on his basics for a month and after that I started taking a weekly test sesions. After few months he started to improve gradually. Now after teaching him for about a year I can proudly say that he has improved alot. The most important thing was he managed to communicate all the difficullties he was facing. He was quite capable and patient. I had a sincere desire to help him reach to its full potential. So I managed to do that. We had a very good honest relationship of a student and a teacher. I loved teaching him as a tutor. Now having an experience of one year teaching I can read students quite well. I look forward to work as an online tutor who could help students in solving their all sort of difficulties, problems and queries.
4.90+
29+ Reviews
43+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Thermodynamics questions
-
In a refrigerator, 2.00 mol of an ideal monatomic gas are taken through the cycle shown in the figure. The temperature at point A is 800.0 K. (a) What are the temperature and pressure at point D? (b)...
-
The temperature of 2.00 mol of an ideal monatomic gas is raised 15.0 K at constant volume. What are (a) The work W done by the gas, (b) The energy transferred as heat Q, (c) The change Eint in the...
-
The temperature of 2.00 mol of an ideal monatomic gas is raised 15.0 K in an adiabatic process. What are? (a) The work W done by the gas, (b) The energy transferred as heat Q, (c) The change Eint in...
-
In Exercises find the second derivative of the function. f(x) = 8 (x - 2)
-
Hartwell Company manufactures one product, it does not maintain any beginning or ending inventories, and its uses a standard cost system. Its predetermined overhead rate includes $1,760,000 of fixed...
-
It is specified that a certain type of iron should contain .85 g of silicon per 100 g of iron (.85%). The silicon content of each of 25 randomly selected iron specimens was determined, and the...
-
What are some theories of gender stratification?
-
Dell Computer Company produces computers. The following data and descriptions are from the company's annual report ($ in millions): Assume that Dell uses the periodic inventory system. Suppose a...
-
George bought the following amounts of Stock A over the years: Note: Loss amounts should be indicated with a minus sign. Stock A Stock A Date Purchased 11/21/1996 Number of Shares 1,150 Adjusted...
-
Calculate the potential difference between points a and b in Figure P28.54 and identify which point is at the higher potential. 4.00 V 2.00 4.00 T120 V 10.0
-
Two moles of an ideal monatomic gas have an initial pressure P 1 = 2 atm and an initial volume V 1 = 2 L. The gas is taken through the following quasi-static cycle: It is expanded isothermally until...
-
Repeat Problem 67 with the paths AB and CD representing adiabatic processes. B.
-
For the company in the previous problem, suppose fixed assets are $520,000 and sales are projected to grow to $713,000. How much in new fixed assets are required to support this growth in sales?
-
How can you use social media ethically from the perspective of your employer?
-
How does the distance from the audience impact how speakers should gesture?
-
Why are subpoints useful?
-
What are some reasons people tend to avoid eye contact when speaking?
-
When might one give a special occasion speech?
-
What does the term healthcare finance mean?
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
If there is a fixed percent uncertainty in LASIK reshaping of the cornea, why would you expect those people with the greatest correction to have a poorer chance of normal distant vision after the...
-
A typical coal-fired power plant generates 1000 MW of usable power at an overall thermal efficiency of 40%. (a) What is the rate of heat input to the plant? (b) The plant bums anthracite coal, which...
-
Automotive Thermodynamics A Volkswagen Passat has a six-cylinder Otto cycle engine with compression ratio r = 10.6. The diameter of each cylinder, called the bore of the engine, is 82.5 mm. The...
-
An air conditioner operates on 800 W of power and has a performance coefficient of 2.80 with a room temperature of 21.0oC and an outside temperature of 35.0oC. (a) Calculate the rate of heat removal...
-
7. An electric charge Q is distributed uniformly throughout a nonconducting sphere of radius Ro with a uniform density, p = Q/{(4/3)R}. Use Gauss's Law to determine (a) (5pts) the electric field,...
-
6.(5pts) A total charge Q is distributed uniformly over a ring of radius R. We found an expression for the value of the electric field on the axis of the ring, E(x), a distance x from the center of...
-
A steel cable that weighs 8 lb/ft is used to pull a 500 lb block of concrete from the ground to the top of a 120 ft tall building. Let x be the distance, in feet, from the block to the TOP of the...

Study smarter with the SolutionInn App


