At what position would you expect electrophilic substitution to occur in each of the followingsubstances? (a) (b)
Question:
At what position would you expect electrophilic substitution to occur in each of the followingsubstances?
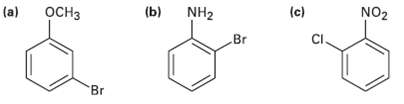
Transcribed Image Text:
(a) (b) (c) оснз NH2 Br NO2 CI. Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a OCH3 OCH 3 OCH3 E Br Br Br E Both groups are orthopara directors and direct substitution ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
At what position would you expect to observe IR absorptions for the following molecules?
-
In each of the following instances, would you expect a positive, negative, or zero correlation? (a) Number of salespersons and total dollar sales for real estate firms. (b) Total payroll and percent...
-
What can you say about the series Σ an in each of the following cases? (b) lim an (a) lim 0.8 an+1 (c) lim
-
The number of claims filed each week with Security Insurance Company has a mean of 700 and a standard deviation of 250. Calculate the probability that the number of claims this week will be: (a)...
-
In 2016, Geoff incurred $900,000 of mine and exploration expenditures. He elects to deduct the expenditures as quickly as the tax law allows for regular income tax purposes. a. How will Geoff's...
-
U.S. defense budget outlays, 19621981. In order to explain the U.S. defense budget, you are asked to consider the following model: Y t = β 1 + β 2 X 2t + β 3 X 3t...
-
For each of the following, calculate the \(t\)-test for independent means. a. X 17.00, SX 1 X 2 = 1.17 - b. X 1 65.56, - sX 1 X 2 = 2.88 X 2 11.00, X-2=60.92, c. X 1 137.73, X-2=114.09, SX 1 X 2 =...
-
How do the ratings of TV and Internet services compare? The file Telecom contains the rating of 14 different providers. (Data extracted from Ratings: TV, Phone, and Internet Services, Consumer...
-
Dave borrowed $600 on January 1, 2022. The bank charged him a $8.00 service charge, and interest was $47.20. If Dave paid the $600 in 12 equal monthly payments, what was the APR? Note: Do not round...
-
The partnership of Dolla, Earl, and Festus was dissolved on January 1, 2014. The balance sheet at that date is shown below: In January, $34,000 of the accounts receivable was collected, and an...
-
Draw resonance structures for the intermediates from reaction of an electrophile at the ortho, Meta and Para positions of nitrobenzene. Which intermediates are most stable?
-
Show the major product(s) from reaction of the following substances with (i) CH3CH2Cl, AlCl3 and (ii) HNO3,H2SO4 (b) (a)
-
As a team, select an industry to analyze. Using your web browser, each team member should access the annual report or 10-K for one publicly traded company in the industry, with each member selecting...
-
Discuss the pros and cons of using measures of leverage to assess whether a company faces financial distress?
-
Explain how you took cultural factors and diversity into account when making the assessment and recommending interventions.
-
As an individual, reflect on and discuss the importance of diversity in teams and the relationship of diversity to teams spread across a large area. What sort of cultural considerations should you...
-
Describe the differences between managerial and financial accounting. Describe the differences as they relate to the users of the information, the purpose of the information, and the time frame (past...
-
How the process of mixing investment assets among different types of investment products, such as cash, fixed income, equity, real estate, derivatives, private investments, and collectibles, is...
-
Name some items that would ordinarily be subject to confiscation under a statute proscribing possession of drug paraphernalia.
-
A 6-lb shell moving with a velocity ?? v0k explodes at point D into three fragments which hit the vertical wall at the points indicated. Fragments A, B, and C hit the wall 0.010 s, 0.018 s, and 0.012...
-
Derive the equations necessary to expand Illustration 6.4-1 to include the thermodynamic state variables internal energy, Gibbs energy, and Helmholtz energy. Illustration 6.4-1 Making of a...
-
Show the major products of these eliminationreactions: CI + NaOCH,CH3 Ph + NaOCH,CH, ELOH ELOH a) Ph Br d) Ph + NaOH CH;OH c) N(CH3)3 OTs
-
When 2-methyl-2-propanol is treated with sulfuric acid, 2-methylpropene is formed. Show all of the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of...
-
Show all of the steps in the mechanism for this reaction, what other products would you expect to beformed? CH3 Br CH3 . CH,O -CH,CH CH.C- C=C CH,CH3 CH3 .
-
According to the Google, Inc. Privacy Policy Litigation, on what date did Google announce their new universal privacy policy?
-
alcolm and Brianna were married on December 15, 2022. They spent the remainder of the tax year in Paris, France. Malcolm is a lifelong resident of North Carolina. Brianna was a lifelong resident of...
-
You work for the strategy department at Bank of America. They have developed a new retirement investment product and are trying to figure out the most cost effective way to encourage customers to...

Study smarter with the SolutionInn App


