Show the major product(s) from reaction of the following substances with (i) CH3CH2Cl, AlCl3 and (ii) HNO3,H2SO4
Question:
Show the major product(s) from reaction of the following substances with (i) CH3CH2Cl, AlCl3 and (ii) HNO3,H2SO4
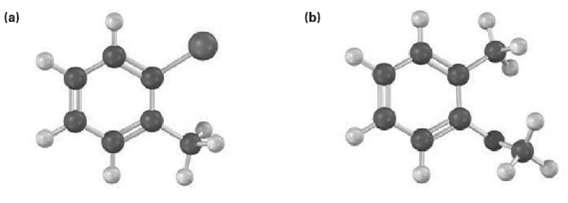
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a Br CH3 CH3 1 CH3CHCl AICI 3 OCH3 ii HNO3 HSO4 i CH3CHCl AICI 3 ii HNO3 HS...View the full answer

Answered By

Abigael martinez
I have been a tutor for over 3 years and have had the opportunity to work with students of all ages and backgrounds. I have a strong belief that all students have the ability to learn and succeed if given the right tools and support. I am patient and adaptable, and I take the time to get to know each student's individual learning style in order to best support their needs. I am confident in my ability to help students improve their grades and reach their academic goals.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures ofall. H (a) . (b) CH3D2H
-
Show the major product for the following transformation: COOEt H3O" C10H100 heat (A) (B) (D) (C)
-
The structures of some common crystalline substances follow. Show that the net composition of each unit cell corresponds to the correct formula of each substance. CI Na Oa cs CI Ti
-
Suppose x = e y . Compute the value of y, given the following values of x: (a) x = 2 (b) x = 3 (c) x ~ 1.5 (d) x 3 (e) x = .5 (f) x = .002 (g) x = 10 (h) x = 1
-
In March 2016, Helen Carlon acquired used equipment for her business at a cost of $300,000. The equipment is five-year property for depreciation purposes. a. If Helen depreciates the equipment using...
-
The XTown manufactures an entry-level hoverboard, HOV-X. Pursuing a cost-leadership strategy, the company has tried to improve quality and reduce costs. As a result of the actions taken over the last...
-
In performing tests of balances on inventory balances, the auditor should recognize that the following potential errors may occur or exist: 1. All inventory items are not counted or tagged. 2....
-
Weaver Companys predetermined overhead rate is $18.00 per direct labor-hour and its direct labor wage rate is $12.00 per hour. The following information pertains to Job A-200: Direct materials...
-
A 37 kg shop sign hangs at one end of a uniform beam of length 1.3 m and 75 kg that is hinged on a front wall of the shop. The beam is supported by another cable as shown in the figure below that...
-
A large manufacturing firm is concerned about lost production (i.e., production capability that was not utilized for a variety of reasons). One of the causes of such lost production was identified as...
-
At what position would you expect electrophilic substitution to occur in each of the followingsubstances? (a) (b) (c) NH2 Br NO2 CI. Br
-
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose amechanism. FC. F3C. "CH-CH NO2 "CH-CH NO2 yfluorfen
-
Judges in Bernalillo County in New Mexico were ranked for their DWI conviction rates and their recidivism rates, where recidivism refers to a subsequent DWI arrest for a person previously charged...
-
The company has provisioned $US1.7 billion ($2.4 billion) to fund Renova's work to December. Discuss the difference between provision and contingent liability from an accounting perspective.
-
An insulated beaker with negligible mass contains liquid water with a mass of 0.240 kg and a temperature of 65.1 C. 9 Part A How much ice at a temperature of -10.8 C must be dropped into the water so...
-
Two electrons in a vacuum exert force of F = 3 . 2 3 E - 0 8 N on each other. They are then moved so that they are separated by x = 5 . 1 times their original distance. How far apart, in meters, were...
-
Two charges q 1 and q 2 have a total charge of 1 0 mu or micro CC . When they are separated by 4 . 8 m , the force exerted by one charge on the other has a magnitude of 8 mN . Find q 1 and q 2 if...
-
Tweaks for newsletter New ideas for newsletter add on. For company newsletter not for email newsletter How can we implement it in a company of solar power energy?
-
An adjustable-rate preferred share is currently selling at a dividend yield of 9%. Assume that the dividend rate on the stock is adjusted once a year and that it is currently paying an annual...
-
1. What is the semi-annually compounded interest rate if $200 accumulates to $318.77 in eight years? Answer in percentage with two decimal places. 2. What is the quarterly compounded interest rate if...
-
Ethylene at 30 bar and 100C passes through a heaterexpander and emerges at 20 bar and 150C. There is no flow of work into or out of the heater-expander, but heat is supplied. Assuming that ethylene...
-
Show the products of thesereactions: CH3 Br CH,CH,OH CH,OH a) Ph- -Br b) Ph CH; CH, Br . CH;OH Br d) ELOH Ph
-
Show the products of thesereactions: Br Br O NaOH NAOH b) PHCHCHCCH3 a)
-
Show the substitution and/or elimination products for these reactions. Explain which mechanism are occurring and which product you expect to be the majorone. OTs a) CH,CH,CHCH, + CH,CH,CO CH,CH,CO,H...
-
3) Two masses are connected with a thin string over a massless, frictionless pulley. If the 12.0 kg box descends 14.0 m in 8.00 seconds after being released from rest, draw two free body diagrams and...
-
For this problem, consider a model of the hydrogen atom where the "atom" is an electron confined to be in a one-dimensional box. Take the length of the box, L, to be twice the Bohr radius ao. The...
-
A floating ice block is pushed through a displacement a = (23.0 m ) - (22.0 m ), along a straight embankment by rushing water, which exerts a force = (266 N )i (186 (186 N ); on displacement? on the...

Study smarter with the SolutionInn App


