Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The
Question:
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is more stable than the other. The less stable one is rapidly converted Co the more stable one, so it cannot be isolated. On the basis of bond dissociation energies, which of these two isomers is more stable?
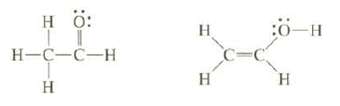
Transcribed Image Text:
H ö: H-C-C-H Т Н н H :ӧ-н H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (20 reviews)
Use Table 21 in the text to calculate total bond energies of compound...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The land-pan-evaporation formula may be used to estimate the evaporation rate from swimming pools. Compare the water loss rate for a pool in Pasadena, California, exposed to air at 80F and 30 percent...
-
What methods can be used to estimate the energy requirements of a job?
-
The formula t = d/4 can be used to estimate the time, t, in seconds it takes for an object dropped to travel d feet. (a) Estimate the time it takes for an object to drop 100 ft. (b) Estimate the time...
-
Governments assert that their safety standards for food imports are important to ensure that their citizens not be harmed by unsafe foods. Comment on how such a concern may be a nontariff barrier.
-
Zap is a retailer that specialises in electrical goods. It is a division of a large retail company, data relate to the most recent year of operations:...
-
A state health department conducted an experiment to evaluate the reliability of assessing the level of contamination of e. coli in three food sources, meat, fruit, and vegetables. There are four...
-
Using the following data, test the question of whether an equal number of boys and girls participate in soccer at the elementary level at the .01 level of significance. What's your conclusion? Gender...
-
Calculate the net present value (NPV) for the following 20-year projects. Comment on the acceptability of each. Assume that the firm has an opportunity cost of 14%. a. Initial investment is $10,000;...
-
You need to implement the following for the users in the marketing department: Open a customer relationship management (CRM) popup when the users receive a Teams call. Ensure that SIP devices can be...
-
Draw ER Diagram: Modern School of higher education has around 2000 students and three departments. The students belong to various departments in the school. Every department has a Head of Department...
-
Because of two hydrogen bonds, carboxylic acids show a very strong attractive force between two molecules that persists even in the gas phase. Show this hydrogen bonding between two carboxylic acid...
-
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is...
-
If the data meet the assumptions of the parametric test, why is it preferable to use the parametric test rather than the nonparametric alternative?
-
__________ companies often centralize staff functions to support all business units. ___________ companies often decentralize staff functions, dedicating them to each business or product...
-
In March 2010 Fernandez and Sicilia signed an LOI with an enterprise valuation of MX $389 million (US $30 million). They valued the company with a number of measures, including comparable logistics...
-
Cristol Company produces sheets. The master budget shows the following standards information and indicates the company expected to produce and sell 27,000 units for the year. Direct Materials 3 yards...
-
Value investor Mohnish Pabrai ( 2 0 0 7 ) wrote the Dhandho Investor: The Low - Risk Value Method to High Returns. Pabrai sums up the Dhandho approach as heads, I win; tails, I dont lose much!...
-
World Travel leases airplanes to airline companies around the world. World Travel is contemplating buying 2 5 additional airplanes for its fleet. It is confident that this purchase will not affect...
-
Give three examples to support the statement "longrange interaction forms the underlying basis for most life processes and technology.
-
Consider a game of poker being played with a standard 52-card deck (four suits, each of which has 13 different denominations of cards). At a certain point in the game, six cards have been exposed. Of...
-
If f(x) = x 2 and g(x) = x 3, find the composite functions f g and g f.
-
The herbicide acifluorfen can be prepared by a route that begins with reaction between a phenol and an aryl fluoride. Propose a mechanism. NO2 CO2CH3 NO2 co,CH3 NO2 .CO2H DMSO HO F3C F3C CI FC...
-
The red fox (Vulpes) uses a chemical communication system based on scent marks in urine. Recent work has shown one component of fox urine to be a sulfide. Mass spectral analysis of the pure...
-
Anethole, C10H12O, a major constituent of the oil of anise, has the 1H NMR spectrum shown. On oxidation with Na2Cr2O7, Anethole yields p-methoxybenzoic acid. What is the structure of Anethole? Assign...
-
1. Write a reflection on advantages and disadvantages of a major decision you took a. A self-evaluation the advantages and disadvantages (on a scale from 1-10). b. A justification for your...
-
BBF315/05 Risk Management May 2023 Assignment 2 Question 1 (a) Risk management is sometimes described as the process of identifying and evaluating the trade-off between risk and expected return, and...
-
A 1.0000x10 -11 m wavelength x-ray photon strikes an atom in an example of Compton scattering. The scattered photon has a wavelength of 1.0301x10 -11 m at an angle of 29 o from the initial direction....

Study smarter with the SolutionInn App


