Calculate the pKa for these compounds. a) HCOH (K 1.75 X 104) b) CHCH3 (K = 10-50)
Question:
Calculate the pKa for these compounds.
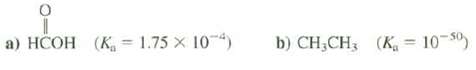
Transcribed Image Text:
a) HCOH (K 1.75 X 104) b) CH₂CH3 (K = 10-50)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
p K a...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using the pKa values of analogous compounds in Table 3.1, predict which would be the stronger base. (a) (b) (c) (d) or (CHa),Cor O: HO or
-
Compounds like CCl2F2 are known as chlorofluorocarbons, or CFCs. These compounds were once widely used as refrigerants but are now being replaced by compounds that are believed to be less harmful to...
-
The pKa of protonated acetone is about -7.5 and the pKa of protonated hydroxylamine is 6.0. a. In its reaction with hydroxylamine at pH = 4.5 (Figure 18.2), what fraction of acetone will be present...
-
Q1-Mutual funds provide the following for their shareholders. A. diversification B. professional management C. record keeping and administration D. all of these options
-
Office Magic Ltd manufactures ergonomic office equipment. The standard cost for material and labour is $190 per unit. This includes 8 kilograms of direct material at a standard cost of $5 per...
-
Orange growers sell $15 billion of their crop to orange juice processors and $6 billion of their crop to supermarkets. The orange juice processors sell their orange juice to supermarkets for $18...
-
A 1.2-lb kite with an area of \(6 \mathrm{ft}^{2}\) flies in a \(20-\mathrm{ft} / \mathrm{s}\) wind such that the weightless string makes an angle of \(55^{\circ}\) relative to the horizontal. If the...
-
Jones, wishing to retire from a business enterprise that he had been conducting for a number of years, sold all of the assets of the business to Jackson Corp. Included in the assets were a number of...
-
MACRO In September 2007, country A presents the following data in its population survey active (in thousands of people) total population 7710 Population aged 15 and over outside institutions 6326...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Which of these species can behave as a Lewis base? a) CH0CH,CH, CH3 d) CH3NH + b) CH3CHCH3 0: I.. e) CHCOH c) CH3NH
-
Calculate the Ka for these compounds. a) HC=CH (pK = 25) b) HC N (pK = 9.31)
-
1. Why is Pablo reluctant to turn over the reins to Jos? Include a discussion of Figure 15.1 in your answer. 2. Cite and discuss two reasons that Pablo should begin thinking about succession...
-
What are the primary categories of a risk?
-
Describe the various court rulings involving PPACA.
-
Why did Congress pass the Health Care Quality Improvement Act of 1986?
-
Describe the common models of managed care organizations.
-
Discuss the purpose and various titles of the Patient Protection and Affordable Care Act of 2010 (PPACA).
-
Graph the U.S. capital-labor ratio since 1960 (use private fixed assets from the Fixed Assets section of the BEA website, www.bea.gov, Table 6.2 as the measure of capital, and civilian employment as...
-
a. What is the cost of borrowing if Amarjit borrows $28 500 and repays it over a four-year period? b. How many shares of each stock would he get if he used the $28 500 and invested equally in all...
-
Prove the identity. sin(/2 + x) = cos x
-
Show how the Wittig reaction might be used to prepare the following alkenes. Identify the alkyl halide and the carbonyl components that would he used. (b) (a)
-
How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following compounds? (a) 2-Pentanol (b) 1-Butanol (c) 1-Phenylcyclohexanol (d) Diphenyl methanol
-
Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing the product with acid. For example, (a) How would you prepare...
-
Respond to this students posting. In your response, you may want to comment on the ethical issues that arise in different career fields or give suggestions for dealing with the ethical questions that...
-
2. A project requires $150 million investment. If you invest today, the present value of future cash flow is worth $180 million. You can wait for one year to observe the demand of the market;...
-
How can a company successfully differentiate their product or service in a highly saturated market, and what are the most effective marketing strategies to communicate this unique value proposition...

Study smarter with the SolutionInn App


