Calculate the total charge due to the charge distributions labeled A, B, C infigure. x=-2 P.=I mC/m
Question:
Calculate the total charge due to the charge distributions labeled A, B, C infigure.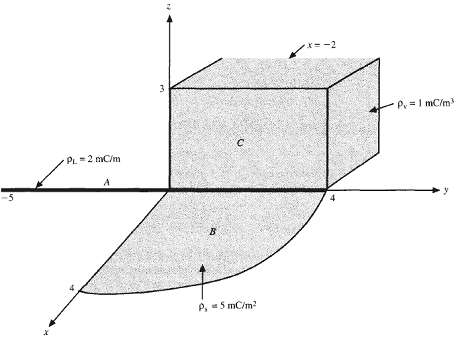
Transcribed Image Text:
x=-2 P.=I mC/m PL 2 mC/m -5 P. 5 mC/m?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Q fpdl p dl 5p 10 mC S ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Electrodynamics questions
-
Due to the charge distribution labeled B in figure,(a) Find E at point (0, 0, 3) if ?S = 5 mC/m2.(b) Find E at point (0, 0, 3) if ?S = 5 sin ? mC/m2. x=-2 P.=I mC/m PL 2 mC/m -5 P. 5 mC/m?
-
A total charge of 7.50 106 C is distributed on two different small metal spheres. When the spheres are 6.00 cm apart, they each feel a repulsive force of 20.0 N. How much charge is on each sphere?
-
A point charge Q = +4.60 C is held fixed at the origin. A second point charge q = + 1.20 C with mass of 2.80 X 10-4 kg is placed on the x-rods, 0.250 m from the origin. (a) What is the electric...
-
Write out the equation for the following chemical reaction and balance it. Solid potassium chlorate, KCIO3, decomposes to form solid potassium chloride and diatomic oxygen gas. What is the...
-
Why might Fed policymakers, in turn, experience difficulties determining which of the public's inflation expectations are the best signals of inflationary pressures in the economy?
-
Data Set 12 Passive and Active Smoke includes cotinine levels measured in a group of smokers (n = 40, x = 172.48 ng>mL, s = 119.50 ng>mL) and a group of nonsmokers not exposed to tobacco smoke (n =...
-
Acetic acid is esterified in the liquid phase with ethanol at \(373.15 \mathrm{~K}\) and \(1 \mathrm{~atm}\) pressure to produce ethyl acetate and water according to the reaction \[ \mathrm{CH}_{3}...
-
Petrocal, Inc., refines gasoline and sells it through its own Petrocal gas stations. On the basis of market research, Petrocal determines that 60% of the overall gasoline market consists of service-...
-
From the intensity function for two point sources, the location and intensity of the peak can easily be calculated. It is very different for the intensity function of a single finite-width slit in...
-
Crystal Cleaners dry cleans industrial clothing. The following excerpt from its PPE Subledger shows the component details regarding the dry cleaning equipment: Calculate depreciation on the dry...
-
Determine the total charge (a) On line 0 < x < 5 m if pz = 12x mC/m (b) On the cylinder p = 3,0 < z < < 4 m if es = pz2 nC/m? (c) Within the sphere r = 4 m if p, %3D C/m r sin 0 10
-
Find E at (5, 0, 0) due to charge distribution labeled A infigure. x=-2 P.=I mC/m PL 2 mC/m -5 P. 5 mC/m?
-
Look in the Yellow Pages or contact your local chamber of commerce or local chapter of CMA-Canada to find the names of manufacturing companies in your area. Call or make an appointment to meet with...
-
We have suggested that group members adapt to the different rules for communication, or code switch, when the context of the social system changes. Competent communicators are aware that different...
-
State the complement of each of the following events: (a) Drawing a spade from a full deck of cards (b) Inflation of less than 5 % per year (c) GNP growth of more than 4 % per year
-
Using Bloomberg, what is the current price and yield on a 30year Treasury bond? Is the bond selling above or below par? Is the yield to maturity (current yield) above or below the coupon rate? What...
-
How do you describe yourself?
-
Describe an outcome you would like to achieve.
-
Design a phase equalizer for the elliptic filter of Exercise 6.6 with the same order as the filter. Exercise 6.6 Design highpass Butterworth, Chebyshev, and elliptic filters that satisfy the...
-
Marc Company assembles products from a group of interconnecting parts. The company produces some of the parts and buys some from outside vendors. The vendor for Part X has just increased its price by...
-
According to the experimental results in Eq. 7.33, is the addition of bromine to cis-2-butene a syn- or an anti-addition? H3C-CH=CH-CH3 cis trans Br CHCl2 Br Br 1 T H3C-CH-CH-CH3 racemate meso (7.33)
-
Explain the reasons for the projected increase in the U.S. budget deficit in coming years.
-
Why is the current path of fiscal policy not sustainable?
-
List the options for making fiscal policy sustainable.
-
Balance, September 1 Checks BANK STATEMENT Deposits Other Balance $ 1,940 September 7 NSF check $ 90 1,850 September 11 $ 2,910 4,760 September 12 #101 $ 770 3,990 September 17 #102 1,640 2,350...
-
A large hospital required nurses to work 80 hours over two-week (14 day) pay periods. In addition, nurses were also considered to be "on-call" for an additional 20 hours during the pay period. While...
-
Describe the history of the HIPAA, explaining why it is essential. Consider the following questions in your response: Why is understanding the history of the HIPAA law essential in decision making...

Study smarter with the SolutionInn App


