Cardiolipins arc a group of lipids found in heart muscles. What products would be formed if all
Question:
Cardiolipins arc a group of lipids found in heart muscles. What products would be formed if all ester bonds, including phosphates, were saponified by treatment with aqueousNaOH?
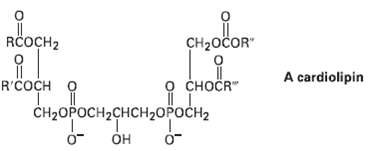
Transcribed Image Text:
RČOCH2 CH2OČOR" A cardiolipin R'COCH O O CHOČR" Ttanganda CH2ОPОСН2СHCH2OPОСH2 OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
Saponification of a Cardiolipins yields 4 different c...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The plasmalogens are a group of lipids found in nerve and muscle cells. How do plasmalogens differ fromfats? CH2OCH=CHR CR A plasmalogen H2OR"
-
What products would be obtained when acetophenone reacts under each of the following conditions? (a) (b) (c) (d) (e) (f) (g) Acetophenone HNO H2S04 CBH NHNH2, HA NaBH CH2OH (1) CBHMgBr (2) NHC...
-
What products would be isolated from urine when dogs are fed (a) phenylheptanoic acid and (b) phenyloctanoic acid? How does this experiment, originally performed by Knoop, shed light on the process...
-
Upland Co.'s inventory records showed the following data accounted for in a perpetual inventory system. Date Units Unit Cost June 1 Inventory 1,250 $8.00 June 3 Purchases 2,500 8.40 June 7 Sales (at...
-
How does a firm acquire other companies to increase the number of innovations it produces and improve its capability to produce innovations? Discuss.
-
If a small part of this magnet loses its superconducting properties and the resistance of the magnet wire suddenly rises from 0 to a constant 0.005 , how much time will it take for the current to...
-
Consider a correctly specified regression model with $p$ terms, including the intercept. Make the usual assumptions about $\varepsilon$. Prove that \[\sum_{i=1}^{n}...
-
The bookkeeper for Willingham Corporation made these errors in journalizing and posting. 1. A credit posting of $400 to Accounts Receivable was omitted. 2. A debit posting of $750 for Prepaid...
-
You wish to test if two population means are equal to each other, against an alternative that they are different. You may assume equal variances. You have two samples n1-12 and n2-12. What is the...
-
Imagine that you are Magna's new corporate controller and answer the following: 1. Describe Magna's strategy in terms of how it competes for customers. 2. Based on Magna's strategy and the data...
-
What products would you obtain from hydrolysis of a plasmalogens (Problem 27.16) with aqueous NaOH with H3O+?
-
Stearolic acid, C18H32O2 yields stearic acid on catalytic hydrogenation and undergoes oxidative cleavage with ozone to yield nonanoic acid and nonanedioic acid. What is the structure of Stearolic...
-
Find f (t) if F(s) is given by expression F(s) = s(s + 1) / (s = 2)2 (s + 3)
-
Jess is a paralegal who received his license four months ago. He works for a senior paralegal named Loralei who is representing the defendant on a complicated collection matter. Jess and Lorelai...
-
a chart showing the relevant time periods of the offering process including the pre-filing or "quiet period", "the waiting period" and the post effective period. Make a quick and crude list of...
-
The SEC has provided certain safe harbors from the prohibition on both Section 5's restrictions on pre-filing offers and the definition of a prospectus for companies not yet public? Name and describe...
-
Please answer TWO discussions 1. Hello Class, Here in the united states, the duty to act responsibly and reasonably is something one should apply when driving whether you're on s highway or not. One...
-
Chapter 13 - Contracts: https://bookshelf.vitalsource.com/reader/books/9781260512700/epubcfi/6/52[%3Bvnd.vst.idref%3Dchapter13]!/12/2/2/2[page_323]/1:6[e%203%2C23] Chapter 16 - Capacity and Legality:...
-
In what situation is the risk of a project likely to match that of the overall firm?
-
Solve each equation or inequality. |6x8-4 = 0
-
What mass of HClO 4 must be present in 0.500 L of solution to obtain a solution with each pH value? a. pH = 2.50 b. pH = 1.50 c. pH = 0.50
-
The structure of poly(vinyl alcohol) is shown below. This polymer cannot be made by polymerizing its monomer. Why not? How could poly(vinyl alcohol) be prepared from poly(vinyl acetate)? --(CH2CH,-...
-
Explain why sulfonation of polystyrene occurs in the positions shown in the ion-exchange resin formula.
-
Although propylene can be polymerized with a free-radical initiator, the molecular weight of the polymer is never very high by this method because chain transfer from the methyl group of propylene...
-
The highest WACC is 11%, the baseline WACC is 7.4% and lowest WACCS is 2.8%. To derive a WACC that better accounts for extreme scenarios, you decide to assign the following probability weights: High-...
-
Give 2 examples of the ff: Statutory law Regulatory law Common law Include the title of each example and explain why you chose these examples ( 1 to 2 sentences ) . Note: Write your reference ( s )
-
The Adams family includes a financially well-informed couple, both aged 36, and two children aged 4 and 6. The family is financially sound but suffered badly during the tech meltdown in 2000. The...

Study smarter with the SolutionInn App


