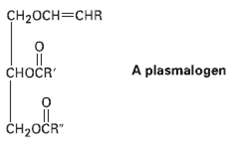
The plasmalogens are a group of lipids found in nerve and muscle cells. How do plasmalogens differ
Question:
The plasmalogens are a group of lipids found in nerve and muscle cells. How do plasmalogens differ fromfats?
Transcribed Image Text:
CH2OCH=CHR сноCR A plasmalogen ČH2OČR"
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
Fats and plasmalogens are both esters of a glycerol mol...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Cardiolipins arc a group of lipids found in heart muscles. What products would be formed if all ester bonds, including phosphates, were saponified by treatment with aqueousNaOH? ROCH2 CH2OOR" A...
-
The cyclitols are a group of carbocyclic sugar derivatives having the general formulation 1, 2, 3, 4, 5, 6-cyclohexanehexol. How many stereo isomeric cyclitols are possible? Draw them in their chair...
-
Polychlorinated biphenyls (PCBs) are a group of synthetic oil-like chemicals that were at one time widely used as insulation in electrical equipment and were discharged into rivers. They were...
-
Ice Cool produces two different models of air conditioners. The activities, costs, and cost drivers associated with the production processes follow. Budgeted Activity Usage 7,600 Activity Machining...
-
1. Is there evidence, in this video about Sheryl Connelly that strategic entrepreneurship exists at Ford? 2. Does Sheryl Connelly set the stage for corporate entrepreneurship? 3. What entrepreneurial...
-
A long, straight conductor passes through the center of a metal ring, perpendicular to its plane. If the current in the conductor increases, is a current induced in the ring? Explain.
-
Consider the hypotheses for the general linear model, which are of the form \[H_{0}: \mathbf{T} \beta=\mathbf{c}, \quad H_{1}: \mathbf{T} \beta eq \mathbf{c}\] where $\mathbf{T}$ is a $q \times p$...
-
Eminence Corporation makes rocking chairs. The chairs move through two departments during production. Lumber is cut into chair parts in the cutting department, which transfers the parts to the...
-
K Daniel Hair Stylists' adjusted trial balance follows. Prepare Daniel's income statement for the year ended December 31, 2024. (Click on the icon to view the adjusted trial balance.) Net Income...
-
Questions 4. What would be a reasonable estimate of total cost for January of Year 2? 5. With only a 5 percent risk of being wrong, would you accept without investigation a January of Year 2 actual...
-
Spermaceti, a fragrant substance from sperm whales, was much used in cosmetics until it was banned in 1976 to protect the whales from extinction. Chemically, spermaceti is cetyl palmitate, the ester...
-
What products would you obtain from hydrolysis of a plasmalogens (Problem 27.16) with aqueous NaOH with H3O+?
-
If one firm raises the costs of another firm by bidding against it for its inputs, that is not an externality by our definition. But, if a firm raises the costs of another firm by polluting the...
-
Kate sold her home for $300,000. She owned and lived in the home for 8 years. The home originally cost Kate $200,000. How much of the gain, if any, i included in her gross income ?
-
Kingston Manufacturing employs the weighted-average method in its process costing system. The Casting Department is the third department in its production sequence. Below is a summary of the...
-
At the end of November, the HIM department has a YTD payroll budget of $350,000. The actual YTD amount is $375,000 because a new assistant director was hired earlier in the year than budgeted and at...
-
Harry's Pottery Equipment Company Stock pays a $4.30 dividend. The required rate of return by the investors is 8.0%. Based on the preferred stock (no growth) model, what is the price of the stock?
-
The Ashton Manufacturing Company operates under a job-order costing system. The data from the past month's activities is summarized as follows: Starting balance in Work in Process: $36,000 Raw...
-
Repeat Problem 6.6-1, but with the process transfer function given by \[ G(s)=\frac{20}{s(3 s+1)} \] Solve for the unit-step responses for \(0 \leq t \leq 1.0 \mathrm{~s}\). Problem 6.6-1 Consider...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
What volume of a concentrated HCl solution, which is 36.0% HCl by mass and has a density of 1.179 g/mL, should be used to make 5.00 L of an HCl solution with a pH of 1.8?
-
How many monomer units will be present in the next product if the diester-diol and diester diacid in eq. 14.26 react? Draw the structure of the product.
-
What product will mainly be formed if a diacid is treated with a large excess of a diol? If a diol is treated with a large excess of a diacid? These reactions represent two extremes of what can...
-
Kodel is a polyester with the following structure: From what two monomers is it made? -O-CH CH2-O
-
4. The water in Earth's atmosphere blocks most of the infrared waves coming from space. In order to observe light of this wavelength, the Kuiper Airborne Observatory has been developed. The...
-
What amount of gain should Pharoah recognize on the exchange assuming lack of commercial substance?
-
If R1=7 Q, R2=3 Q, R3=9 Q, V= 12 V, and V2=8 V. Find the current (in A) that pass through R. R R R V V 2 +

Study smarter with the SolutionInn App


