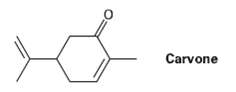
Carvone is the major constituent of spearmint oil. What products would you expect from reaction of carvone
Question:
Carvone is the major constituent of spearmint oil. What products would you expect from reaction of carvone with the following reagents?
(a) (CH3)2Cu ? Li +, then H3O +?
(b) LiAlH4, then H3O
(c) CH3NH2?
(d) C6H5MgBr, then H3O+
(e) H2/Pd?
(f) CrO3, H3O+
(g) (C6H5)3P+C?HCH3?
(h) HOCH2CH2OH, HCI
Transcribed Image Text:
Carvone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a d g O HO ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from reaction of 1-methylcyclohexene with the following reagents? (a) Aqueous acidic KMnO4 (b) O3, followed by Zn, CH3CO2H
-
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures ofall. H (a) . (b) CH3D2H
-
What products would you expect from reaction of estradiol (Problem 27.44) with the following reagents? (a) NaH, then CH3I (b) CH3COCI, pyridine (c) Br2, FeBr3 (d) Pyridinium chlorochromate in CH2Cl2
-
What are the side effects of the drugs that are used for treatment of Multiple Sclerosis attacks? Are cataracts a result of steroid use? Is osteoporosis a complication of Multiple Sclerosis?
-
How does Goodwill use controls to make informed decisions about its resources?
-
On a test with 20 questions, a student must answer 8 of the first 12 questions and 4 of the last 8. In how many ways can this be done?
-
What is IV&V and how is it applied?
-
Consider the following all-integer linear program: Max 1x1 + 1x2 s.t. 4x1 + 6x2 22 1x1 + 5x2 15 2x1 + 1x2 9 x1, x2 0 and integer a. Graph the constraints for this problem. Use dots to indicate...
-
3. Given Right Triangle. Side a is 4.5 cm. Side c is 6.5 cm. Find another side and angles of the triangle. Sketch the diagram. 4. Given Force 34.0 N SE direction under angle 55 above horizontal. Find...
-
Step-by-Step Painting prepares and packages paint products. Step-by-Step has two departments: (1) Blending and (2) Packaging. Direct materials are added at the beginning of the blending process...
-
How would you synthesize the following substances from benzaldehyde and any other reagentsneeded? (c) (b) (a) CH- CH2
-
The SN2 reaction of (dihromomethyl) benzene, C6H5CHBr2, with NaOH yields benzaldehyde rather than (dihydroxymethyl) benzene, C6H5CH (0H)2 Explain.
-
Juan is helping his mother rearrange the living room furniture. Juan pushes on the armchair with a force of 30 N directed at an angle of 15 above a horizontal line while his mother pushes with a...
-
9. Ehsan Corporation has 800 million shares outstanding trading at $40 per share. The company wants to raise $2 billion and are considering using rights offering. A) If the offering requires 10...
-
Mando Enterprises is expected to pay a dividend of $2.50 a year from today. Annual dividends are expected to grow by 10% in year 2, by 7% in year 3, and thereafter by 4.50% per year. Stockholders...
-
Pepi's Pizza Parlour has a cost of debt of 6% and a cost of equity of 10%. The firm has 100,000 shares of common stock outstanding at a book price of $20 a share. Shares are now trading at $22. The...
-
Suppose that a fund manager has a portfolio of stocks currently worth (in total) 100,000 and that on the market there are a call and a put option contracts available on the same assets of the...
-
what extent does the reciprocal exchange of feedback and constructive critique within the mentorship dyad serve as a catalyst for personal and professional metamorphosis, engendering a culture of...
-
While we have focused on the Lagrangian and action for point particles in this chapter, it is possible to formulate the Schdinger equation itself through the principle of least action with a...
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
The formal charges on the O atoms in the ion [ONO] + is (a) -2; (b) -1; (c) 0; (d) +1.
-
In the presence of sodium ethoxide the following transformation occurs. Explain. (1) NaOEt (2) HCI OEt OEt
-
Thymine is one of the heterocyclic bases found in DNA. Starting with ethyl propanoate and using any other needed reagents, show how you might synthesize thymine. CH3 Thymine
-
Predict the products from each of the following aldol reactions. (a) (b) (c) (d) (e) H NaOH H20 H H NaOH H2O o'H H HOEN NaOH H2O H NaOH H2O
-
es Hart, Attorney at Law, experienced the following transactions in Year 1, the first year of operations: 1. Accepted $16,600 on April 1, Year 1, as a retainer for services to be performed evenly...
-
Dahlia Corporation has a current accounts receivable balance of $439,516. Credit sales for the year just ended were $5,503,810. a. What is the receivables turnover? Note: Do not round intermediate...
-
Why does the organizational structure hold political significance? Provide an in-depth analysis of this concept using examples from both academic literature and real-world instances. Additionally,...

Study smarter with the SolutionInn App


