What products would you expect from reaction of the following alkenes with NBS? If more than one
Question:
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures ofall.
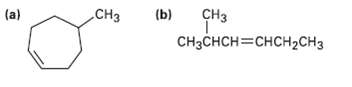
Transcribed Image Text:
сHз (a) .СНз (b) CнзснсH3Dснсн2СHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a CH3 5Methyl cycloheptene NBS CC14 heat CH3 Br 3Bromo5methyl cycloheptene Br C...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from reaction of 1-methylcyclohexene with the following reagents? (a) Aqueous acidic KMnO4 (b) O3, followed by Zn, CH3CO2H
-
What products would you expect from each of the following reactions? (a) (b) (c) t-BuOK CHBr CH2Zn(Cu) diethyl ether
-
What product would you expect from reaction of 1 equivalent of methanol with a cyclic anhydride, such as phthalic anhydride (1, 2-benzenedicarboxylic anhydride)? What is the fate of the second ?half?...
-
A saving bond earns a variable rate of interest that can change six months, with compounding done monthly. The initial rate was 6.8% in early 2015. If that rate continues unchanged for the 3 years of...
-
Consider the stylized example in Figure 7.13. Repeat the forecasts using the additive model with trend, using the same smoothing constants. FIGURE 7.13 Stylized Model with Trend and Cyclic...
-
Given the circuit in Fig. 3.53, calculate the currents i1 through i4? Figure 3.53 2A 0 IA
-
Fab and Fast Ltd buys and sells motor vehicle accessories. The firms estimated sales and expenses for the first 4 months of 2025 are shown below. Actual sales for December 2024 were $900000 and...
-
Suppose that Carrier Lumber Ltd, a Canadian forest products company, sells lumber to Home Depot. In return, Home Depot will pay Carrier US$1,000,000 in 90 days. At a current exchange rate of...
-
The Super Bowl Indicator Theory suggests that the stock market will have a positive year if the National Football Conference team, or a team with an NFC origin, wins. If the American Football...
-
The following information from St. Paul Supply, Inc. is provided for 2017 and 2016: 12/31/17 12/31/16 12% Cumulative preferred stock, $20 par $200,000 $200,000 Common stock, $5 par 40,000 28,000...
-
The major product of the reaction of methylenecyclohexane with N-bromo-succinimide is 1-(bromomethyl) cyclohexane. Explain CH2Br CH2 NBS Major product
-
How would you prepare the following alkyl halides from the correspondingalcohols? CH (a) CI (b) Br CH CHH2CH CH (c) CI H (d) BrCH2CH2CH2CHCH CHCH2H2cH3 CH
-
Figure shows an overhead view of a corridor with a plane mirror M mounted at one end. A burglar B sneaks along the corridor directly toward the center of the mirror. It d = 3.0 m, how far from the...
-
The following questions is based on strategic leadership. What is the opinion of employees on strategic leadership? How does strategic leadership approach used by the leader provide employees...
-
Rizio Company purchases a machine for $9,900, terms 2/10, n/60, FOB shipping point. Rizio paid within the discount period and took the $198 discount. Transportation costs of $224 were paid by Rizio....
-
Introduction: Provide a concise, professional introduction explaining the purpose of your analysis to your executive. Horizontal and Vertical Analysis: In this section, you will conduct horizontal...
-
Consider the matrix A = 1 1 1 1) 2 1 0 2 and the vector b = (i) Find the null space of A: N(A) (ii) Find a basis matrix for the null space of A: Z(A) (iii) Represent a feasible solution for Ax= b in...
-
Were the public relations tactics of the food industry ethical? Why or why not? I am always amazed when I watch a documentary about something I think I already know, then learn so much more. The role...
-
A multiple-choice question on a statistics quiz has possible correct answers of a, b, c, d, e. What is the probability that false is the correct answer?
-
How is use of the word consistent helpful in fraud reports?
-
Which metal is most easily oxidized? (a) Na (b) Cr (c) Au
-
The heat of combustion of cis-1, 2-dimethylcyclopropane is larger than that of the trans isomer. Which isomer is more stable? Use drawings to explain this difference in stability.
-
Trans-1, 2-Dimethylcyclobutane is more stable than cis-1, 2-dimethylcyclobutane, but cis-1, 3-dimethylcyclobutane is more stable than trans-1, 3-dimethylcyclobutane. Use drawings to explain these...
-
Name the following alkanes and haloalkanes. When two or more substituents are present, list them in alphabetical order. (a) (b) (c) (d) CH CH CH-CH CHCH Br CH CH CH CH CH CH CH CH, CH,CH(CH3)....
-
Select a company you are familiar with and detail one short-term and one longer-term strategy, then discuss how the role of human resource development can provide valuable advice and counsel in the...
-
Sunspot Beverages, Limited, of Fiji uses the weighted-average method of process costing. It makes blended tropical fruit drinks in two stages. Fruit juices are extracted from fresh fruits and then...
-
How does TPM intersect with Lean Manufacturing principles? Discuss the synergies between TPM and Lean practices, such as continuous improvement (Kaizen) and value stream mapping, in reducing waste...

Study smarter with the SolutionInn App


