We saw in Section 17.4 that ketones react with NaBH 4 to yield alcohols. We?ll also see
Question:
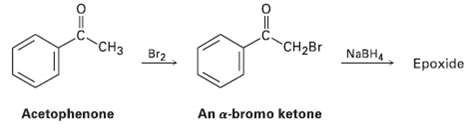
We saw in Section 17.4 that ketones react with NaBH4 to yield alcohols. We?ll also see in Section 22.3 that ketones react with Br2 to yield a-bromo ketones. Perhaps surprisingly, treatment with NaBH4 of the a-bromo ketone from acetophenone yields an epoxide rather than a bromo alcohol. Show the structure of the epoxide, and explain its formation.
Transcribed Image Text:
CH2BR NABH Ерохide CH3 Br2 An a-bromo ketone Acetophenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
H CHBr addition of hydride to the ketone CH Br H di...View the full answer

Answered By

Abigael martinez
I have been a tutor for over 3 years and have had the opportunity to work with students of all ages and backgrounds. I have a strong belief that all students have the ability to learn and succeed if given the right tools and support. I am patient and adaptable, and I take the time to get to know each student's individual learning style in order to best support their needs. I am confident in my ability to help students improve their grades and reach their academic goals.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
We saw in Section 11.5 that, to find the energies of the bonding and antibonding orbitals of a heteronuclear diatomic molecule, we need to solve the secular determinant where aA et aB and we have...
-
We saw in Section 25.6 that DCC can be used to form a peptide bond. We explored the mechanism, and we saw that DCC activates the COOH moiety so that it readily undergoes nucleophilic acyl...
-
We saw in Section 10.1 that the undiscounted risk-neutral expected stock price equals the forward price. We will verify this using the binomial tree in Figure 11.4. a. Using S = $100, r = 0.08, and ...
-
Joan has been a full-time director of Sunnybank Pursuits Ltd (a trading company) since 2001 and has owned 10% of the company's ordinary shares since 2002. She retired in February 2021 and gave all of...
-
Explain the differences between implementation controls, strategic surveillance, and special alert controls. Give an example of each.
-
What factors might explain differences between co-worker and supervisor ratings?
-
Throughout the US presidential election of 2016, polls gave regular updates on the sample proportion supporting each candidate and the margin of error for the estimates. This attempt to predict the...
-
Simon Company determines that its goodwill is impaired. It finds that its implied goodwill is $360,000 and its recorded goodwill is $400,000. The fair value of its identifiable assets is $1,450,000....
-
what ways do modern enterprises leverage iterative methodologies such as Six Sigma and Lean Management to systematically identify inefficiencies, streamline processes, and cultivate a culture of...
-
Exhibit 1: Prices and projected annual sales volumes for Sony PlayStation 3 and Microsoft Xbox 360 Elite Exhibit 2: Sony PlayStation 3 Production Costs per unit in dollars Exhibit 3: Microsoft Xbox...
-
Aldehydes and ketones undergo acid-catalyzed reaction with alcohols to yield hemiacetals, compounds that have one alcohol-like oxygen and one ether-like oxygen bonded to the same carbon. Further...
-
Give IUPAC name for the following compounds: (c) , (b) (a) CHCH,CHCHCH CH CH2CH2CH3 -CH CH C (e) . (f) (d) Br Br
-
Prepare a table with the following headings for a monthly bank reconciliation dated September 30. Indicate whether each item should be added to or subtracted from the book or bank balance and whether...
-
What is meant by 'defence of due diligence'? In the workplace, who would need to be aware of the concept of due diligence?
-
What is not a place where due diligence might happen?
-
how would you explain the significance and application of Residency and Family Law Act and Assignments and Preferences Act/ Fraudulent Conveyances Act when conveying interests in 'fee simple'?
-
Outline and critically discuss the viability of the grounds of divorce recognised under our Namibian Law. NB: Strictly Namibian and South African Law ONLY.
-
Due diligence is the process of investigating the full and complete implications of buying a business. Drawing from your knowledge of due diligence, suggest processes and methods that could be used...
-
Why do you think the U.S. Supreme Court has never held that the Fifth Amendment requirement of indictment by a grand jury applies to the states as well as to the federal government?
-
Anne is employed by Bradley Contracting Company. Bradley has a $1.3 million contract to build a small group of outbuildings in a national park. Anne alleges that Bradley Contracting has discriminated...
-
A 1.5 kW heater is to be used to heat a room with dimensions 3.5 m 5.0 m 3.0 m. There are no heat losses from the room, but the room is not airtight, so the pressure in the room is always...
-
Using a three-dimensional formula, show the direction of the dipole moment of CH3OH. Write (+ and (- signs next to the appropriate atoms.
-
Trichloromethane (CHCl3, also called chloroform) has a larger dipole moment than CFCl3. Use three-dimensional structures and bond moments to explain this fact.
-
Indicate the direction of the important bond moments in each of the following compounds (neglect C-H bonds). You should also give the direction of the net dipole moment for the molecule. If there is...
-
MR Green is one of the resident for who you take care in a nursing home .He is recovering from a stroke and has a pressure ulcer . In addition he is disoriented and confuse . However he can feed...
-
What does the accountability of prescriptive theory? b. What does the accountability of capture theory? c. What does the accountability of normative accounting theory? d, What does the accountability...
-
Describe the connections between leader accountability and how leaders hold people accountable for their actions? sustainability through accountability.

Study smarter with the SolutionInn App


