Give IUPAC name for the following compounds: (c) , (b) (a) CHCH,CHCHCH CH
Question:
Give IUPAC name for the following compounds:
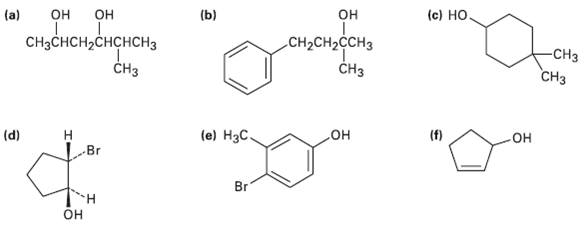
Transcribed Image Text:
(c) НО, он (b) нснсненс он он (a) CHзснCH,CHCHCHЗ CHз CH2CH2ĊCH3 -CHз CHз Cнз (e) Нзс. (f) (d) Н Br он но Br Он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
The parent chain must contain the hydroxyl group and the hydroxyl ...View the full answer

Answered By

Vineet Kumar Yadav
I am a biotech engineer and cleared jee exam 2 times and also i am a math tutor. topper comunity , chegg India, vedantu doubt expert( solving doubt for iit jee student on the online doubt solving app in live chat with student)
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC name for the following compounds (reddish brown =Br): (a) (b) (c)
-
Give IUPAC name for the followingcompounds: (c) NC. CH 2 () (a) CHCH2CH2CH CH2 CH () CH CH2CO2H (d) (e) CHH2H2H2CH3 CHCN CH (g) Br (h) CN BRCH2CHCH2CH2CO2H
-
Give an IUPAC name for the following compounds: q, b.CH,CH2CHCH, c' .CH,HCHCH, CH3 CH,--C-CH3 CH3 ,
-
On 1 May 2006, Nigel acquired a 30-year lease for 20,000. He assigned the lease on 1 November 2020 for 75,000. Kay purchased land in February 2012 for 20,000. She sold one-third of the land for...
-
Select a business whose strategy is familiar to you. Identify what you think are the key premises of the strategy. Then select the key indicators that you would use to monitor each of these premises.
-
Early this year, a taxpayer was clearing dry brush from behind his Malibu home in California. He became frustrated with how long it was taking using his clippers. He decided instead to light the...
-
CD Real Estate specializes in residential real estate services in the state of California. To complement the experience and local market knowledge of its licensed realtors, CD Real Estate wants to...
-
Presented here are liability items for Azarian Inc. at December 31, 2012. Prepare the liabilities section of Azarian??s balance sheet. Accounts payable Notes payable (due May 1, 2013) Bonds payable...
-
1) Calculate the magnitude of the electric field generated (8.4x10^0) m from an electron, giving your answer in N/C to 2 significant figures. Magnitudes must be positive. 2) A non-conducting...
-
(a) If a diffraction grating has a resolution of 10 4 , is it possible to distinguish two spectral lines with wavelengths of 10.00 and 10.01 m? (b) With a resolution of 10 4 , how close in...
-
We saw in Section 17.4 that ketones react with NaBH 4 to yield alcohols. We?ll also see in Section 22.3 that ketones react with Br 2 to yield a-bromo ketones. Perhaps surprisingly, treatment with...
-
Draw structures corresponding to the following IUPAC name: (a) (Z)-2-Ethyl-2-buten-1-o1 (b) 3-Cyclohexen-1-o1 (c) trans-3-Chlorocycloheptanol (d) 1, 4-Pentanediol (e) 2, 6-Dimethylphenol (f)...
-
Show that the probabilities in a Cox, Ross, and Rubinstein binomial tree are negative when the condition in footnote 8 holds.
-
What three practical concepts have you learned from listening to the Borrowed Future Episode 1 Podcast" what no one told you about student loan"?
-
Summarize this information below. Introduction: The Croc Your World Enterprise (CYW) company founded by the charismatic and socially responsible businessman, Mr. Victor Diego. CYW's mission is to...
-
Discuss TWO (2) factors used by underwriters in the selection and classification of risks in order to achieve equity among policyholders using your own word.
-
Briefly explain the legal framework that regulates public sector financial Operations in Kenya by giving a minimum of five examples.
-
A tank contains mercury up to a height of 0.3 m over which water stands to a depth of 1 m and oil of specific gravity 0.8 stands to a depth of 0.5 m over water. Determine the pressure the interfaces...
-
Identify the research scenario, including the relevant independent and dependent variables. Develop a hypothetical research scenario that would necessitate the use of a Regression-Discontinuity...
-
Consider the combustion of methanol below. If 64 grams of methanol reacts with 160 grams of oxygen, what is the CHANGE in volume at STP. 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(1) The volume decreases by...
-
Repeat the calculation in Problem 3.25, but now assume that the second cylinder, instead of being evacuated, is filled with nitrogen gas at 20 bar and 160 K. Problem 3.25 A 0.01-m 3 cylinder...
-
Write structural formulas for all of the alkenes with (a) The formula C2H2Br2 and (b) The formula C2Br2Cl2. In each instance designate compounds that are cis-trans isomers of each other. Predict the...
-
Write structures for all compounds with molecular formula C4H6O that would not be expected to exhibit infrared absorption in the 3200-3550-cm-1 and 1620-1780-cm-1 regions.
-
Add curved arrows to the following reactions to indicate the flow of electrons for all of the bond-forming and bond-breaking steps. (a) (b) H.
-
1) Explain how a Preschool program can help to bring a sense of belonging for the child? 2) It is important for a program to provide physical environments that are age-appropriate and that support...
-
Your company decided to organize a special dinner to appreciate all the employees and it has been proposed that the dinner to be held in the middle of December 2021. You have been appointed as the...
-
Select a two-dimensional image to analyze that is either a work of art (photograph, illustration, or painting) or an advertisement. What is the FTC analysis?Use the FTC palette to break down the...

Study smarter with the SolutionInn App


