Choline, a component of the phospholipids in cell membranes, can be prepared by SN2 reaction of trimethyl
Question:
Choline, a component of the phospholipids in cell membranes, can be prepared by SN2 reaction of trimethyl amine with ethylene oxide. Show the structure of choline, and propose a mechanism for thereaction.
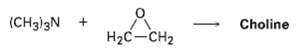
Transcribed Image Text:
(CHд])3N + Choline H2C-CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (19 reviews)
The reaction of trimethylamin...View the full answer

Answered By

Shadrack Mulunga
I am a Biochemistry by profession. However, I have explored different fields of study. My quest to explore new fields has helped me gain new knowledge and skills in Business, clinical psychology, sociology, organizational behavior and general management, and Project Management. I count my expertise in Project management, in particular, creation of Work Break Down Structure (WBS) and use of Microsoft Project software as one of my greatest achievement in Freelancing industry. I have helped thousands of BSC and MSC students to complete their projects on time and cost-effectively using the MS Project tool. Generally, I find happiness in translating my knowledge and expertise to success of my clients. So far, i have helped thousands of students to not only complete their projects in time but also receive high grades in their respective courses. Quality and timely delivery are the two key aspects that define my work. All those who hired my services always come back for my service. If you hire my services today, you will surely return for more. Try me today!
5.00+
154+ Reviews
289+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Ethers can often be prepared by S N 2 reaction of alkoxide ions, RO ? , with alkyl halides. Suppose you wanted to prepare cyclohexyl methyl ether. Which of the two possible routes shown below would...
-
A compound known as Hagemann's ester can be prepared by treating a mixture of formaldehyde and ethyl acetoacetate first with base and then with acid and heat. Write the structure for the product of...
-
Choline (Sec. 11.11) can be prepared by the reaction of trimethylamine with ethylene oxide. Write an equation for the reaction, and show its mechanism.
-
1. An auditor selected a product maintained in the finished goods Warehouse. The auditor counted the product and compare this amount what the amount in the finished goods Perpetual inventory...
-
Finding job openings that align perfectly with your professional interests is wonderful, but it doesn't always happen. Sometimes you have to widen your search and go after whatever opportunities...
-
Standard addition graph. Students performed an experiment like that in Figure 5-7 in which each flask contained 25.00 mL of serum, varying additions of 2.640 M NaCl standard, and a total volume of...
-
Which of the following procedures is not appropriate for identifying laws and regulations that management may have overlooked? a. Discussing laws and regulations with the entity's chief financial...
-
James Lawsons Bed and Breakfast, in a small historic Mississippi town, must decide how to subdivide (remodel) the large old home that will become its inn. There are three alternatives: Option A would...
-
Explain the basic workings of a refrigerator. Discuss the refrigerator in terms of thermodynamic terminology.
-
Etep lives permanently in New Hampshire and works at UMass Lowell. She teaches in-person three days a week (Monday/Wednesday/Friday) and works from home (in New Hampshire) on the other four days. She...
-
Chlorophyll, heme, vitamin B12, and a host of other substances are bio-synthesized from porphohilinogen (PBG), which is itself formed from condensation of two molecules of 5-aminolevulinate. The two...
-
Cyclopentamine is an amphetamine-like central nervous system stimulant. Propose a synthesis of Cyclopentamine from materials of five carbons orless. CH3 -CH2CHNHCH3 Cyclopentamine
-
Identify and describe the four modes of moisture movement into and through building assemblies.
-
How do management assertions in financial reporting contribute to the overall quality and reliability of financial statements? In what ways can errors or misstatements in these assertions impact the...
-
On January 1, Year 4, Dean purchased all of Kay's $10 par, voting common stock for $600,000. On that date, the fair values of Kay's assets and liabilities equaled their carrying amounts of $660,000...
-
On January 1, Year 3, JoJo Co purchased 15,000 of the voting shares of Bo Inc. for $20 per share. Bo has 150,000 voting shares outstanding. Management elected to use FVOCI. Bo paid dividends of...
-
Sean and Connie are considering starting a manufacturing business. Each will contributed $200,000 and own 50% of the business. They believe their net taxable income from ordinary business activities...
-
Ms. Smith, owner of Smith Dental Supply Company, and has been in business for one year. She applied for a $150,000 loan from her bank. Assume she is successful in getting the loan. How will this loan...
-
JDLs current ratio is: (a) 1.0 to 1. (b) 0.7 to 1. (c) 1.5 to 1. (d) 2.4 to 1. JDL Corporation Selected Financial Data, December 31, 2024 Current assets $150,000 Current liabilities 100,000...
-
(a) Use integration by parts to show that (b) If f and g are inverse functions and f' is continuous, prove that (c) In the case where f and t are positive functions and b > a > 0, draw a diagram to...
-
What is a general characteristic of a Lewis acid? Of a Lewis base?
-
Will 2-hexyne react with sodium amide? Explain.
-
For the following compounds, write structural formulas and IUPAC names for all possible isomers having the indicated number of multiple bonds: a. C4H6 (one triple bond) b. C5H10 (one double bond) c....
-
Name the following compounds by the IUPAC system: a. CH3CH=C(CH2CH2CH3)2 b. (CH3)2CHCH"CHCH3 c. g. CH3-C-C-CH-CH, h. k.
-
Complete Telecom Solution's worksheet for the month ended November 3 0 , 2 0 2 4 . How much was net incoke for November? Requirements 1. 2. Complete Telecom Solution's worksheet for the month ended...
-
Question: Part One: Memo Write an analysis of the impact of tax laws and incentives as demonstrated in this current years tax return. Specifically, you must address the following: Explain the...
-
From the following ledger balances, prepare a trial balance for the Cheyenne Corp. at June 3 0 , 2 0 2 2 . All account balances are normal.Accounts Payable $ 1 0 , 0 0 0 , Cash $ 7 , 4 0 0 , Common...

Study smarter with the SolutionInn App


