Color and absorption spectra. Color Plate 15 shows colored solutions and their spectra. From Table 17-1, predict
Question:
Color and absorption spectra. Color Plate 15 shows colored solutions and their spectra. From Table 17-1, predict the color of each solution from the wavelength of maximum absorption. Do observed colors agree with predicted colors?
Table 17-1
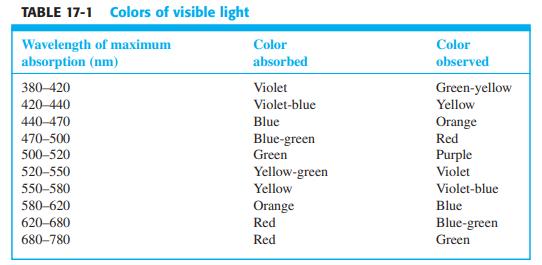
TABLE 17-1 Colors of visible light Wavelength of maximum absorption (nm) Color Color absorbed observed 380-420 Violet Green-yellow 420-440 Violet-blue Yellow 440-470 Blue Orange 470-500 500-520 Red Blue-green Green Purple 520-550 Yellow-green Violet 550-580 Yellow Violet-blue 580-620 Orange Blue 620-680 Red Blue-green 680-780 Red Green
Step by Step Answer:
Curve A B D E F Absorption peak nm 760 ...View the full answer



Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Chemical Engineering questions
-
The photograph of upconversion in Color Plate 19 shows total internal reflection of the blue ray inside the cuvet. The angle of incidence of the blue ray on the wall of the cuvet is 55. We estimate...
-
The solutions shown here each have an absorption spectrum with a single absorption peak like that shown in Figure 23.26. What color does each solution absorb most strongly? [Section 23.5]
-
Predict the positions of the absorption bands in the IR spectra for the carbonyl groups of thesecompounds. HO. b) CH,CH-CHCH (p
-
One major concern about the future is that water scarcity will grow, particularly in arid regions where precipitation levels may be reduced by climate change. Will our institutions provide for an...
-
In a recent newspaper release, the president of Downs Company asserted that something has to be done about depreciation. The president said, Depreciation does not come close to accumulating the cash...
-
A deep pool of water is initially at \(4^{\circ} \mathrm{C}\) and suddenly the surface is exposed to a freezing front at \(-4{ }^{\circ} \mathrm{C}\) and remains so exposed for a long time. Find the...
-
Broken rotor bars are not a common fault. What are two main causes of broken rotor bars?
-
McLaughlin borrowed $1,000 from Adler, who, apprehensive about McLaughlins ability to pay, demanded security. McLaughlin indorsed and delivered to Adler a negotiable promissory note executed by...
-
The following transactions occurred during August: (a) Invested cash in the business, $30,000. (b) Purchased a used van for cash, $8,000. (c) Purchased plumbing equipment on account, $4,000. (d)...
-
You are a team of financial consultants hired to assist the client, Mr. Bansal. Read the case individually and jot down your notes. Discuss the case with your group. Evaluate the proposed project for...
-
Characteristic orange light produced by sodium in a flame is due to an intense emission called the sodium D line, which is actually a doublet, with wavelengths (measured in vacuum) of 589.157 88 and...
-
This problem can be worked by calculator or with the spreadsheet in Figure 18-5. Consider compounds X and Y in the example labeled "Analysis of a Mixture, Using Equations 18-6" in Section 18-1. Find...
-
A \(1.00-\mathrm{mol}\) sample of an ideal diatomic gas in which the gas particles both translate and rotate is initially at \(600 \mathrm{~K}\). Energy is then added thermally to the sample until...
-
If you have swam one lap in a swimming pool, that means you have swam from one end to the other and back. In 4 . 5 h , a swimmer makes exactly 3 2 . 5 laps in a swimming pool that is 2 2 m long....
-
Explain the relationship between Cuba and African Liberation Movements.
-
What were some successes and failures of both Capitalist and Socialist regimes in Africa from 1960-1985?
-
How does multiple characters in William Goldings Lord of the Flies drastically change as they devolve from humanity into savagery?
-
What does "liberty" or "freedom" mean in the context of William Bradford's "Of Plymouth Plantation"?
-
Calculate the slope of the relationship in Problem 4 between 2,253 and 3,372 theatres. LEGO Tops the Box Office Movie The LEGO Movie About Last Night RoboCop The Monuments Men Theatres (number) 3,775...
-
In a system with light damping (c < cc), the period of vibration is commonly defined as the time interval d = 2/d corresponding to two successive points where the displacement-time curve touches one...
-
Consider the following reaction: 2 NO(g) + 2 H2(g) -- N2(g) + 2 H2O(g) (a) The rate law for this reaction is first order in H2 and second order in NO. Write the rate law. (b) If the rate constant for...
-
Consider the following reaction: CH3Br(aq) + OH (aq) -- CH3OH(aq) + Br(aq) The rate law for this reaction is first order in CH3Br and first order in OH. When [CH3Br] is 5.0 10-3 M and [OH] 0.050 M,...
-
The reaction between ethyl bromide (C2H5Br) and hydroxide ion in ethyl alcohol at 330 K, C2H5Br(alc) + OH (alc) -- C2H5OH(I) + Br(aIc), is first order each in ethyl bromide and hydroxide ion. When...
-
2.1 Explain by means of drawings how the Successive-Approximation conversion process takes place when receiving an analog voltage input. Use the KEEP/RESET method. Use 5 V input. Vref= 8 V (10) 2.2...
-
Assume that we have a Ridge regression problem with only one predictor, and the true model is linear without an intercept, i.e. Y = X + e. Assume that we have In samples, (xi, y), (x2, Y2), . . .,...
-
Please write a program that calculates the final score of multiple students using different weight of exams and assign their final grade using the following criteria: If the average grade is 90 or...

Study smarter with the SolutionInn App


