Predict the positions of the absorption bands in the IR spectra for the carbonyl groups of thesecompounds.
Question:
Predict the positions of the absorption bands in the IR spectra for the carbonyl groups of thesecompounds.
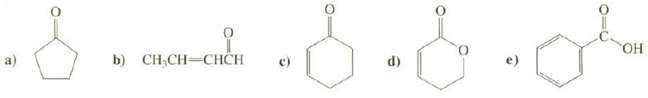
Transcribed Image Text:
HO. b) CH,CH-CHCH (p
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a The carbonyl group of this ketone is part of a five membered ring so the band will be s...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the positions of the major absorption bands in the IR spectra of thesecompounds: CH30 CH-NH, a) CH3CH=CHCCH; c) b) C-H NO, e) CH-CH,C3- d) CH,CH,CH,OH CH3
-
Indicate the positions of the absorption bands and any other noteworthy features in the hydrogen region of the IR spectra of thesecompounds: CH3 ) C,CH b) NH2 d) CH2=CHCH,CH,OH f) CH,CH,CH,CH e)...
-
Predict the key IR absorption bands whose presence would allow each compound in pairs (a), (c), (d), (e), (g), and (i) from Problem 2.46 to be distinguished from each other. In problems 2.46 (a) (c)...
-
On the cost of goods manufactured schedule, depreciation onfactory equipment... A. is not listed because it is not a product cost. B. is not an inventoriable cost. C. is not listed because it is...
-
From experience and/or research, explain what you think are the best practices when training, developing and providing base salaries for expatriates. Describe the differences that people may see in...
-
In Young's two-slit experiment, the first dark fringe above the central bright fringe occurs at an angle of 0.44. What is the ratio of the slit separation, d, to the wavelength of the light, ?
-
Consider a 10-year zero-coupon bond with face value \(\$ 100\). The interest rate is fixed at 5\%. The credit spread for the bond is estimated to be \(1 \%\) (except in part (a)). Calculate: (a) The...
-
Qamar, Inc., did not pay dividends in 2009 or 2010, even though 50,000 shares of its 6.5%, $50 par value cumulative preferred stock were outstanding during those years. The company has 800,000 shares...
-
Calculate Rm (market return from market price index) and Rj (return of share prices). Date PERIOD 1 AORD R company m m Feb-14 5403 0.008 0.3370 Mar-14 5470.8 0.008 0.3354 Apr-14 5473.8 0.008 0.3298...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
The exhaust from a poorly maintained automobile may contain a wide variety of different hydrocarbon pollutants. Why is the 3000 to 2900 cm1 region a good place to monitor the amount of these...
-
Explain how IR spectroscopy could be used to distinguish between thesecompounds: CH,COCH, and COCH,CH, a) CH,CH,CH,CCH, and CH,CH,CH,CH,CH b) c) CH;CH-CHOCH, and CH,CH-CHCH,COH
-
What are the characteristics of an effective performance evaluation interview? How should new and experienced employees be treated differently during the interview?
-
Suppose the monopolist can price discriminate after the 4th unit. That is, the first four units are sold at a price of $27. If they want to sell a 5th unit, they charge $27 from the first four units,...
-
Please complete the rest of the operating budget analysis for CHEWY INC (CHWY via New York Stock Exchange) using your own scholarly source. I included the excel link to the financial statements from...
-
A car was moving at a speed of 7 5 mph for the first 4 miles and then continued move for another 4 miles at a speed of 3 0 mph . What is the average speed of the car?
-
For adult the RDA of the amino acid lysine is 1 2 mg per kg of body weight. How many grams per day should a 7 1 . 0 kg adult receive?
-
FIN 3010 ASSIGNMENT 1 QUESTION ONE (i) "Discounted payback ensures that you don't accept an investment with a negative NPV but it can't stop you from rejecting projects with a positive NPV"...
-
Consider the Odebrecht case and its habit of using bribes and kickbacks to secure project contracts. In what ways is this unethical behavior the fault of the firm, and how is it the fault of the...
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
What likely happened to the four-firm concentration ratio in the airline industry when United and Continental Airlines merged? What likely happened to the eight-firm concentration ratio?
-
1, 3-Cyclopentadiene undergoes thermal polymerization to yield a polymer that has no double bonds in the chain. On strong heating, the polymer breaks down to regenerate cyclopentadiene. Propose a...
-
When styrene, C6H5CH = CH2, is copolymerized in the presence of a few percent p-divinyl benzene, a hard, insoluble, cross-linked polymer is obtained. Show how this cross-linking of polystyrene chains...
-
Poly (ethylene glycol), or Carbowax, is made by anionic polymerization of ethylene oxide using NaOH as catalyst. Propose amechanism. +0-CH2CH2t Polylethylene glycol)
-
Choose from the following topics to create discussion: Humanity and the Natural Environment Housing and Home Furnishings Privacy and Socializing
-
On December 31, 2020, Pearl Company had $1,101,000 of short-term debt in the for January 21, 2021, the company issued 22,800 shares of its common stock for $42 per brokerage fees and other costs of...
-
During your interview process for the position of program manager for Booker, Tee, and How you are asked to explain the relationship between cost, schedule, and performance? what is your answer to...

Study smarter with the SolutionInn App


