Complete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the hybridization of the indicatedatoms.
Question:
Complete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the hybridization of the indicatedatoms.
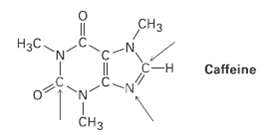
Transcribed Image Text:
CHз H3C. -- Caffeine 0= Cнз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
HC 0 0 CH3 CH3 ...View the full answer

Answered By

Deepak singh
I have completed my graduation from computer science engineering . I have good knowledge about programming language and love to solve many codes of any language. I have been an expert to other platforms too and have good experience of giving solution to the students of subject .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify all nonbonding lone pairs of electron in the following molecules, and tell what geometry you expect for each pf the indicated atoms. (a) The oxygen atom in the dimethyl ether, CH3 ? O ? CH3...
-
The file Caffeine contains the caffeine content (in milli-grams per ounce) for a sample of 26 energy drinks: a. Construct an ordered array. b. Construct a stem- and- leaf display. c. Does the ordered...
-
Identify the expected hybridization state and geometry for the central atom in each of the following compounds: a. b. c. d. e. H. I-z: I-
-
Suppose that Marthas income rises to $42,000 per year, and that she increases her consumption of health care visits by fi ve visits. Using the graphs for Exercise 1, draw the new equilibrium. What is...
-
Dedriea contributes to her wholly owned corporation some tangible personal property that she had used in her sole proprietorship business and depreciated. She had acquired the property for $566,000...
-
You are building inspector in your local community. The emergency manager has asked you to create a report on building construction practices. What should you include in the report?
-
Show that \(\left(\frac{1}{2}, \frac{1}{2} ight)\) is the optimal policy for Example 18.2 . Example 18.2 (Volatility pumping) Suppose there are two assets available for investment. One is a stock...
-
A consulting project involved a credit card bottom feeder (lets call it Credit Wonder). This company bought credit card accounts that were written off as uncollectable debts by major banks. Credit...
-
10. Complete the table below (10pts) Trial Acceleration of gravity "g" m/s 1 9.74054 2 9.98976 3 9.70740. 4 9.70398 5 9.67059 CO 6 9.67177 7 9.70358 8 9.64002 9 9.67337 10 9.57659 11. Take the...
-
(a) Flopro plc make and sell two products A and B, each of which passes through the same automated production operations. The following estimated information is available for period 1: (i) (ii)...
-
Allene (see problem 1.46) is related structurally to carbon dioxide, CO2. Draw a picture showing the orbitals involved in the and bonds CO2, and identify the likely hybridization of carbon.
-
Almost all stable organic species have tetravalent carbon atoms, but species with trivalent carbon atoms also exist. Carbocations are one such class of compounds. (a) How many valence electrons do...
-
Wolvo Company has defective products in inventory. It has the opportunity to either sell, scrap, or rebuild the defective products. Identify several factors Wolvo Company should consider before...
-
With the aid of practical examples, outline ANY Five (5) benefits brought by Information and Communication Technology (ICT) in education from when COVID-19 started to date.
-
OPERATING SYSTEM 1 1.Explain the two main functions that an Operating System performs. 2.Explain the three classes or types of users of a computer system. 3.List and describe five errors that virtual...
-
Operating systems have suitably and rapidly evolved over the years from serial processing operating systems to today's robust multiprogramming, time-sharing and real-time operating systems (i)....
-
Explain the following six (6) operating systems: Batch OS Distributed OS Multitasking OS Network OS Real-OS Mobile OS
-
1.Introduction Background Life Gizmo, inc (LGI) of Boston, MA in the USA have developed the C8F4, a very small device which can be implanted easily and without risks into a person's skull and send a...
-
Is there a valid reason to exempt close family relatives from punishment as accessories after the fact to felonious conduct?
-
Discuss the information available from the following techniques in the analysis of inorganic pigments used in antique oil paintings: (i) Powder X-ray diffraction, (ii) Infrared and Raman...
-
The vant Hoff corollary to the third law of thermodynamics is that whenever two solid forms of a substance are known, the one with the greater specific heat will be the more stable one at higher...
-
Draw structure for the neutral molecule represented by the following model. Explain whether the octet rule is satisfied at each atom of the compound. Draw all of the important resonance structures...
-
Indicate whether each of these species can act as an acid, a base, or both: H +1 a) H-N-H 1 H H e) H-C-O-H H b) H-O-H H: H TI f) H-C-C-C-H H H c) H-C-H H H d):Ci : g) H-O-C-:
-
Show the conjugate acids of these species: a) CH-O-H b) H-O: C) CH,NH,
-
A rhino is charging at you, so decide to sedate it with a tranquilizer gun angled at 40 as shown above. The tranquilizer gun can reach a horizontal distance of 49 m when angled at 45. (Assume ideal...
-
What is the definition of motivation and how can the chef foster an environment of motivation in the kitchen? What methods can a chef implement to motivate the kitchen team? What factors contribute...
-
Article Review Read the article. Link is given below: https://www.projecttimes.com/articles/avoid-the-top-three-real-causes-of-scope-creep.html Post your thoughts on this article, add references if...

Study smarter with the SolutionInn App


