Identify all nonbonding lone pairs of electron in the following molecules, and tell what geometry you expect
Question:
Identify all nonbonding lone pairs of electron in the following molecules, and tell what geometry you expect for each pf the indicated atoms.
(a) The oxygen atom in the dimethyl ether, CH3 ? O ? CH3
(b) The nitrogen atom in trim ethylamine,
(c) The phosphorus atom in the Phosphine, PH3
(d) The sulfur atom in the amino acid methionine,
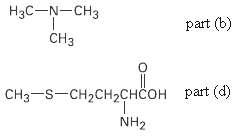
Transcribed Image Text:
Нзс—N—CHз CHз part (b) CHз—S-CH2CH2снсон раrt (d) NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (22 reviews)
a b c HH HI HCNCH H3C HPH H The sp3hybridized oxygen ato...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following molecules and ions are grouped by similar structures. Classify each as aromatic, antiaromatic, or nonaromatic. For the aromatic and antiaromatic species, give the number of pi electrons...
-
Identify the indicated hydrogen?s in the following molecules as pro-R or pro-S: (a) (b) .co2 H3N Alanine (S)-Glyceraldehyde
-
Identify the indicated protons in the following molecules as unrelated, homotopic, enantiotopic, ordiastereotopic: (a) (b) Cysteine
-
What is unrealistic about the domino fault model?
-
What conditions cause the golden parachute arrangement to be penalized?
-
Write a mechanism using sodium ethoxide in ethanol for the epimerization of cis-decalone to trans-decalone. Draw chair conformational structures that show why trans-decalone is more stable than...
-
A convergent-divergent nozzle has an exit throat area ratio of 3.0. It is to be supplied with air. Find: (a) The first, second, and third critical pressure ratios; (b) The exit plane Mach number in...
-
Trevor Moore opened a law office on January 1, 2015. During the first month of operations, the business completed the following transactions: Jan. 1 Moore contributed $ 59,000 cash to the business,...
-
What were some of the socioeconomic conditions in 19th-century America that led the majority of state courts to adopt the legal principle of employment-at-will? How do advocates of the...
-
Circle K farms consumes 3 tons of special feed daily. The feed-a mixture of limestone, corn, and soybean meal-must satisfy the following nutritional requirements: Calcium. At least 0.8% but not more...
-
Draw a line-bond structure for the propyne, CH3C CH; indicate the hydribization of each carbon; and predict a value for each bond angle.
-
Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular formula of each substance: (b) NHCH3 Adrenaline Estrone (a hormone)
-
Preparation method in Exercise 13-12. Use α = 0.05. (a) Apply Fishers LSD method with α = 0.05 and determine which levels of the factor differ. (b) Use the graphical...
-
What is the Recognized Net Capital Gain/Loss from the following transactions? ABC and XYZ stock are publicly traded securities. The vehicle is the taxpayers personal vehicle, used to commute to work...
-
Taylor Inc., a CCPC, received the following non-eligible dividends from taxable Canadian corporations during the current year: Milk Inc. (9% ownership interest) $ 28,000 Juice Co. (40% ownership...
-
How do organizations set up a near monetary record, and what reason does a similar accounting report serve?
-
Broadway Inc. is a CCPC, that is not associated with any other corporations and which has a December 31 year end. The following amounts have been correctly determined for 2023 for Broadway: Active...
-
What are some short term and long term goals a experienced dental owner/office can use to expand their business to generate more revenue? using the information below. Elements Dental facility is...
-
The delay cost in dollars for the sell order of West Commerce shares is: A. \($1\),800. B. \($2\),000. C. \($2\),700. Michelle Wong is a portfolio manager at Star Wealth Management (SWM), an...
-
A routine activity such as pumping gasoline can be related to many of the concepts studied in this text. Suppose that premium unleaded costs $3.75 per gal. Work Exercises in order. Use the...
-
Compute the difference between the pure-component and partial molar enthalpies for both components at 298.15 K and various compositions in each of the following mixtures using the data in Fig....
-
Although carbon?carbon double bonds are shorter than carbon?carbon single bonds, all of the carbon?carbon bonds of benzene are the same length. Explain. H. H C. H H Benzene H H
-
Explain whether or not these molecules are polar (have a dipole moment). (a) CBr 4 (b) NH 3 (c) CH 3 OCH 3 (d) CH 2 C1 2 (e) CO 2
-
For each model, draw a structure that fits the geometry of the molecule. Then explain the geometry at each atom other than the hydrogens.
-
Could Arthur Murray have created language that would have avoided the application of impossibility of performance? What if Parker was not incapac-itated but that it was simply painful to dance after...
-
In the text's case on the county clerk and marriage licenses, whatare the ethical issues involved? What are the conflicting ethical values and whichvalues were maximized/minimized? The Case of the...
-
2 Altira Corporation provides the following information related to its inventory during the month of August 2024: August 1 Inventory on hand-2,000 units%;B cost $5.30 each. August 8 Purchased 8,000...

Study smarter with the SolutionInn App


