Complete the following reactions. (No reaction may be the correct response.) NO2
Question:
Complete the following reactions. (No reaction may be the correct response.)
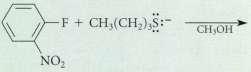
Transcribed Image Text:
NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
The product results ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Complete the following reactions. a. b. c. d. Pt CH3 H CH 13 Fecl Spark CH,C=CH, + 02
-
Complete the following reactions by giving the major organic products. (a) (b) (c) (d) (CH)NH (excess) SOCI CH,CH.CO,H-(excess) Cl-C-CI CH OH (excess O0 O + CH,OH-
-
Complete the following reactions by giving the major organic product(s). (a) (b) (c) +HC(CH,N) P/C 2 25c 1) fuming HNO, H2SO4 2) neutralize with NaOH
-
Craig Industries was in the business of manufacturing charcoal. Craig, the corporation's president, contracted in the name of the corporation to sell the company's plants to Husky Industries. Craig...
-
During 2012 Daveco constructed a new building on land it already owned. In order to finance the construction, on January 1, Daveco took out a $100,000 8% loan, and it already had two other...
-
Yamamoto Iron & Steel is considering setting up a minimill outside Atlanta, Georgia. At present, the company is planning to send a group of executives to the area to talk with local and state...
-
A mutual fund has provided investment yield rates for five consecutive years as follows: Determine \(r_{1}\) and \(r_{2}\), the lag 1 and lag 2 autocorrelation coefficients. Determine \(r_{1}\) and...
-
The following information relates to Karen Weigel Co. for the year 2014. Retained earnings, January 1, 2014 .........$48,000 Dividends during 2014 ............. 5,000 Service revenue ...................
-
6. We have two coins in a box, namely Coin 1 and Coin 2. If we toss Coin 1, its probability of head is 0.9 and probability of tail is 0.1. Coin 2 is a fair coin, i.e., it has 50% chance of head and...
-
Better Homes Products' Canadian operations are organized into two divisions: West and East. West division sells a component that could be used by East Division in making one of the company's...
-
Give a curved-arrow mechanism for the reaction in Eq. 18.85. Be sure to identify the electrophilic species in the reaction and to show how it is formed. Eq. 18.85 CH3 CH 70% H,504 80 C OH + H2O CH3...
-
Within each set, identify the ether that would not readily cleave with concentrated HBr and heat, and explain. Then give the products of ether cleavage and the mechanisms of their formation for the...
-
The Whole Bread Company also allocates fixed manufacturing overhead to products on the basis of standard direct manufacturing labor- hours. For 2012, fixed manufacturing overhead was budgeted at $...
-
A student has an out of district educational placement, paid for by the district. Within the context of the agreement, it allows for the student to continue to participate in district...
-
Discuss how drug laws are inequitably enforcement on minorities and also how we might reform the criminal justice system, in regard to drug laws?
-
Identify the types of information officers can rely on to build reasonable suspicion. Give two examples of each and provide references and in-text citations if possible.
-
In Asheville, NC Buncombe County, what are the beer labeling laws? What styles of beer is produced in the area?
-
Why do people and organizations obey the law? Discuss why you think deterrence is not always effective. Why would individuals/organizations disobey the law when they know there are consequences? Is...
-
Discuss how knowledge of the break-even point and the margin of safety will assist management in its risk assessment of business operations.
-
Transform the while loop from the previous exercise into an equivalent for loop (make sure it produces the same output).
-
Explain how the dipole moments of FCl (0.9 D) and 1Cl (0.7 D) can be so similar.
-
Chlorine is more electronegative than phosphorus. Predict the dipole moment of PCl5.
-
Although carbon?carbon double bonds are shorter than carbon?carbon single bonds, all of the carbon?carbon bonds of benzene are the same length. Explain. H. H C. H H Benzene H H
-
How do emerging technologies such as blockchain and artificial intelligence facilitate stakeholder communication, participation, and feedback mechanisms in modern governance structures ?
-
hen firms kept leases off their balance sheets it tended to a. make the firms appear riskier than they actually were because their stated debt ratios were increased. b. have no effect on either cash...
-
Sunnyside Solar Consultants provided $180,000 of consulting services to Delton Developments on April 14, 2023, on account. Use the PST rates in Exhibit 10.6. Required: Journalize Sunnyside's April 14...

Study smarter with the SolutionInn App


