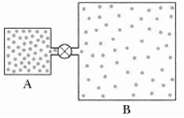
Container A in Figure holds an ideal gas at a pressure of 5.0 x 105 Pa and
Question:
Container A in Figure holds an ideal gas at a pressure of 5.0 x 105 Pa and a temperature of 300 K. It is connected by a thin tube (and a closed valve) to container B, with four times the volume of A. Container B holds the same ideal gas at a pressure of 1.0 x 105 Pa and a temperature of 400 K. The valve is opened to allow the pressures to equalize, but the temperature of each container is maintained. What then is the pressure in the twocontainers?
Transcribed Image Text:
A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
When the valve is closed the number of moles of the gas in cont...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A rigid tank contains an ideal gas at 300 kPa and 600 K. Now half of the gas is withdrawn from the tank and the gas is found at 100 kPa at the end of the process. Determine (a) the final temperature...
-
A rigid tank contains an ideal gas at 40°C that is being stirred by a paddle wheel. The paddle wheel does 200 kJ of work on the ideal gas. It is observed that the temperature of the ideal gas...
-
The gas law for an ideal gas at absolute temperature T (in kelvins), pressure P (in atmospheres), and volume V (in liters) is PV = nRT, where is the number of moles of the gas and R = 0.0821 is the...
-
A survey of recent masters of business administration (MBAs) reveals that their starting salaries follow a normal distribution with mean $48,000 and standard deviation $9,000. Find the probability...
-
Hamlet acquires a 7-year class asset on November 23, 2017, for $100,000. Hamlet does not elect immediate expensing under 179. He does not claim any available additional first-year depreciation....
-
The asking prices (in thousands of dollars) of some four-bedroom homes in Akron, Ohio, are described by the dotplot in Fig. 51. a. A buyer can afford to pay up to $150 thousand. Assuming sellers will...
-
Consider the conditions of Problem 7.36, but now allow for radiation exchange between the surface of the heating element \((\varepsilon=0.8)\) and the walls of the duct, which form a large enclosure...
-
Prior to liquidation, the accounting records of Blazon, Carl, and Dove included the following balances and profit-and-loss percentages: The partnership sold the noncash assets for $74,000, paid the...
-
1. Evaluate the performance management appeals process and its connection to the employee performance. very important no to give short answers very important to give a detailed explanation very...
-
A beam ABCD with a vertical arm CE is supported as a simple beam at A and B (see figure). A cable passes over a small pulley that is attached to the arm at E. One end of the cable is attached to the...
-
An air bubble of volume 20 cm3 is at the bottom of a lake 40 m deep, where the temperature is 4.0oC. The bubble rises to the surface, which is at a temperature of 20oC.Take the temperature of the...
-
Calculate the rms speed of helium atoms at 1000 K. See Appendix F for the molar mass of helium atoms.
-
Subsequent expenditures related to an item of fixed asset should be added to its book value A. Only if it is recommended by the auditors B. Only if it is recommended by the Board of Directors C. Only...
-
What role does memory play in shaping our sense of identity, and how do cognitive processes like autobiographical memory contribute to identity continuity over time?
-
How does the globalization of social structures, characterized by transnational flows of capital, information, and people, reshape traditional boundaries and configurations of power, culture, and...
-
What Self-direct variable annuity contracts are great for investment-savvy clients for what reason?
-
If the percentage change in net income changes faster than the percentage change in revenues, what is happening in the business?
-
What industry standards were used for the basis of the audit? explain all industry standards in detail with examples.
-
On a highway trip, Joseph drives the first 25 miles at 55 mph, and the next 15 miles at 70 mph. What is his average speed for this trip?
-
On July 1, 2011, Flashlight Corporation sold equipment it had recently purchased to an unaffiliated company for $480,000. The equipment had a book value on Flashlights books of $390,000 and a...
-
In 2018, selected automobiles had an average cost of $16,000. The average cost of those same automobiles is now $24,000. What was the rate of increase for these automobiles between the two time...
-
Two adults and a child want to push a wheeled cart in the direction marked x in Fig. 4.36. The two adults Rush with~ horizontal forces F 1 and F 2 as shown in the figure. (a) Find the magnitude and...
-
An oil tanker's engines have broken down, and the wind is blowing the tanker straight toward a reef at a constant speed of 1.5 m/s (Fig. 4.37). When the tanker is 500 m from the reef, the wind dies...
-
A Standing Vertical Jump Basketball player Darrell Griffith is on record as attaining a standing vertical jump of 1.2 m (4 ft). (This means that he moved upward by 1.2 m after his feet left the...
-
Assignment 1 1. Write a program to implement the following formula for a -10 and b = 4; result = (a + 2b - 4ab)^4 // ^4 means to the power of 4 (1 mark) 2. Write a program to implement result = sin...
-
I have most of it done, I just need help fixing some things. import java.util.Scanner; public class Main { private String[] database; private Scanner input; public Main() { database = new...
-
Part 2 - WeatherMonitor Suppose you observe daily temperatures and want to know how hot each day is compared to recent days. That is, you want to: keep track of daily temperature values and know for...

Study smarter with the SolutionInn App


