Convert the following structures to skeletal structures: OH a) CHCH,CH,CHCH,CH, b) HC d) || HC OH CH
Question:
Convert the following structures to skeletal structures:
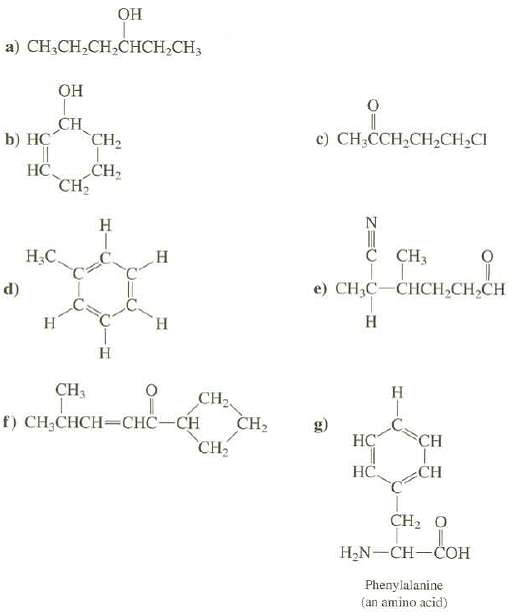
Transcribed Image Text:
OH a) CHỊCH,CH,CHCH,CH, b) HC d) || HC OH CH CH₂ H₂C. H CH3 CH₂ CH₂ H H H H CH₂ f) CH₂CHCH-CHC-CH CH₂ CH₂ 0 11 c) CH3CCH₂CH₂CH₂CI || C CH3 e) CH₂C-CHCH₂CH₂CH H HC HC H CH CH CH, O H₂N-CH-COH Phenylalanine (an amino acid)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
8 a d ...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Convert the following structures into skeletal drawings: (a) (b) H. H. H. Indole 1,3-Pentadiene (d) (c) H. -CI C-CI H. 1,2-Dichlorocyclopentane Benzoquinone U
-
Convert the following condensed structures into skeletal structures (remember that condensed structures show atoms but few, if any, bonds, whereas skeletal structures show bonds but few, if any,...
-
Convert these structures to skeletal structures: T a) H-C-C H H H-C-H C c) CH3CHCCHCH(CH3)2 C=C=C-H b) H H-C-H H H CH3 d) CH,CHCH,NHCH,CH CH,
-
What characteristics do you think Jamie Oliver has that have helped make his various enterprises a success?
-
Contrast the likely differences between the balanced scorecards of a profit seeking company and a non for profit organization?
-
Sendelbach Corporation is a U.S.-based organization with operations throughout the world. One of its subsidiaries is headquartered in Toronto. Although this wholly owned company operates primarily in...
-
Imagine you are working for a company that sells teleconferencing equipment. Draft a value proposition for selling your equipment to a sales manager who has ten salespeople traveling two weeks a...
-
A company issues a $6,000,000, 12%, five-year bond that pays semiannual interest of $360,000 ($6,000,000 12% ), receiving cash of $6,463,304. Journalize the bond issuance.
-
Your t-shirt company sees that shirts of a certain variety have demand function q = -90p + 900 where q is the number of shirts you can sell in one week if you charge $p per shirt. You have fixed...
-
Find the amplitude of the graph of y = cos x
-
Which functional group is present in each of thesecompounds? a) CHCHCHCHOCH, ) CHCH,CH,CO,H Careful: how are the O's bonded to the C? e) CHCOCH, b) d) f) OH 0 CNH, H
-
Convert the following shorthand representations to structures showing all of the atoms, bonds, and unshared electron pairs: d) COOH COH b) OH de c) f) H CN Ni
-
Starlite Company manufactures office products. Last year, it sold 45,000 electric staplers for $10 per unit. The company estimates that this volume represents a 30 percent share of the current...
-
The managers of a grocery store are interested in knowing how many people will shop in their store in a given hour. Suppose they collect data and find that the average number of people who enter the...
-
Write a complete program to create an array of Card object discussed in Sect. 5.6.2. The program should ask a user for the number of cards and using a loop to initialize each card. Perform a couple...
-
Roll a die 25 times and construct a table showing the relative frequency of each of the 6 possible numbers.
-
The SC Johnson Company has a meeting-intensive culture that has spawned its own vocabulary to describe various kinds of meetings. Generals are weekly one-on-one meetings that bosses hold with their...
-
A student measures the acceleration \(A\) of a cart moving down an inclined plane by measuring the time \(T\) that it takes the cart to travel \(1 \mathrm{~m}\) and using the formula \(A=2 / T^{2}\)....
-
PricewaterhouseCoopers (or PwC, as it is known), one of the United States' "Big 4" accounting firms, has had a tax practice in Russia since the time that country changed from Communist rule. One of...
-
In a paragraph of approximately 150-200 words, analyze a film or TV/Streaming Show poster of your choosing by focusing on the ways in which representations in the poster are gendered. Include an...
-
Use the Midpoint Rule with n = 5 to approximate 2 1 J1 X - dx.
-
Benzene and alkyl-substituted benzenes can be hydroxylated by reaction with H2O2 in the presence of an acidic catalyst. What is the structure of the reactive electrophile? Propose a mechanism for...
-
How would you synthesize the following compounds from benzene? Assume that ortho and Para isomers can beseparated. (b) CH2CHCH3 (a) CH Br O2N SO3H
-
You know the mechanism of HBr addition to alkenes, and you know the effects of various substituent groups on aromatic substitution. Use this knowledge to predict which of the following two alkenes...
-
How do advanced scenario planning methodologies, such as probabilistic forecasting, sensitivity analysis, and scenario stress-testing, inform the development of robust, adaptive visions that are...
-
How can leaders sustain momentum and relevance around visionary visions amidst evolving external environments, internal dynamics, and competing priorities, fostering adaptability, resilience, and...
-
What cognitive processes underlie the formulation of visionary narratives, and how can leaders leverage storytelling techniques to evoke emotional resonance, foster buy-in, and drive transformative...

Study smarter with the SolutionInn App


