Which functional group is present in each of thesecompounds? a) CHCHCHCHOCH, ) CHCH,CH,CO,H Careful: how are the
Question:
Which functional group is present in each of thesecompounds?
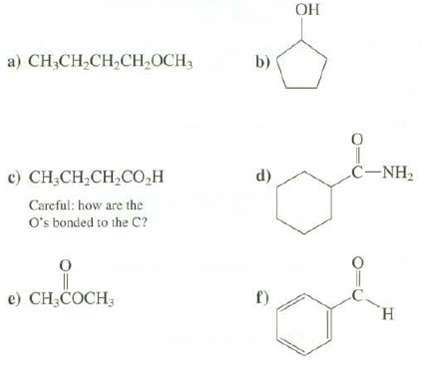
Transcribed Image Text:
a) CH₂CH₂CH₂CH₂OCH, ¢) CHÍCH,CH,CO,H Careful: how are the O's bonded to the C? e) CH₂COCH, b) d) f) OH 0 CNH, H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a Ether b Alcoh...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which transition metal atom is present in each of the following biologically important molecules: (a) Hemoglobin (b) Chlorophylls (c) Siderophores.
-
Name the functional group(s) present in each of the compounds in Problem 2.17.
-
Name the functional group(s) present in each of the compounds in Problem 2.18.
-
Discuss why you would or would not like to work in an organization like this?
-
Explain the purpose of a strategy map and how it relates to a balanced scorecard?
-
Refer to the data from Exercise 12.11. Recall that a model was fit to relate systolic blood pressure to the age and weight of infants. The researcher wants to be able to predict systolic blood...
-
What is the difference between a top-down approach to solving a problem and a bottom-up approach? In what kinds of situations might each be more appropriate?
-
For the past two years, NE Companys best-selling product has been a titanium engine block. Standard direct labor hours per block are 2.0 hours. All direct labor employees are paid $24 per hour....
-
a)Provide an example of a Cross Site Scripting (XSS) attack. Include an explanation of the differences between stored and reflective XSS attacks. b)Provide an example of a SQL Injection attack. Why...
-
Shep Companys records show the following information for the current year. Determine net income (loss) for each of the following separate situations. a. Additional common stock of $3,000 was issued...
-
Which of these compounds would you expect to be more soluble in water? Explain? CHCHCHCHCOH or CH3CHCHCHCHCOH
-
Convert the following structures to skeletal structures: OH a) CHCH,CH,CHCH,CH, b) HC d) || HC OH CH CH HC. H CH3 CH CH H H H H CH f) CHCHCH-CHC-CH CH CH 0 11 c) CH3CCHCHCHCI || C CH3 e) CHC-CHCHCHCH...
-
On December 1, year 1, Peak Advertising (a calendar-year, accrual-basis taxpayer) receives a $24,000 retainer fee for a two-year service contract. a. How much income should Peak report in year 1 for...
-
The 2021 earnings of Sergio, Giox Land Inc./Milan Inc taxes withheld., and the total tax due quarterly after deducting the 5% are shown below. He registered as an 8% taxpayer from the previous years....
-
Springfield Nuclear Energy Inc. bonds are currently trading at $853.67. The bonds have a face value of $1,000, a coupon rate of 4.5% with coupons paidannually, and they mature in 20 years. What is...
-
Explain why it is often more beneficial for people with low incomes to save for retirement in a TFSA instead of an RRSP. For each option below, decide whether a TFSA or RRSP would be more appropriate...
-
There are 3 bonds in the market: Bond A B C Coupon rate (%) 0 9 7 Coupon payments are annual and bid-ask spreads are zero. Projects A B C Maturity 1 2 2 a) What are the prices of the above bonds? (6...
-
1 Which of the following statements must be true of the circuit above? A. The voltage across each capacitor is the same. B. The charge on each capacitor must be the same. C. The voltage across C3...
-
(a) Use Figure \(7.2 a\) to calculate the \(x\) components of the momenta of the two carts at \(t=30,60\), and \(90 \mathrm{~ms}\). (b) What is the \(x\) component of the momentum of the system at...
-
Smiths Family Fashions implemented a balanced scorecard performance measurement system several years ago. Smiths is a locally owned clothing retailer with fashions for men, women, teens, and...
-
Find 1 + x 2 x 5 dx.
-
Propose a mechanism to account for the following reaction: C CH2CI AICI3
-
In the Gatterman-Koch reaction, a formyl group (?CHO) is introduced directly onto a benzene ring. For example, reaction of toluene with CO and HCl in the presence of mixed CuCl/AlCl 3 gives...
-
Treatment of p-tert-butyl phenol with a strong acid such as H2SO4 yields phenol and 2-methyipropene. Propose a mechanism.
-
Two forces act on a 2 7 . 0 kg object floating in space. One force has magnitude of 1 6 . 0 N and acts in the positive y axis direction. The second force has a magnitude of 1 0 . 2 N and acts at an...
-
You have been presented with the basic principles of personal finance, why do you think it is important to study this topic for a finance student? Among the basic principles of personal finance, what...
-
Since the Drug-Free Workplace Act of 1988 many U.S. employers have chosen to conduct drug testing as a pre-employment requirement, random testing of current employees, or a required test after a...

Study smarter with the SolutionInn App


