Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can?t be isolated. Explain, taking the polarity
Question:
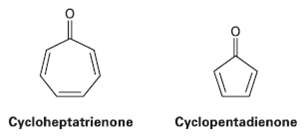
Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can?t be isolated. Explain, taking the polarity of the carbonyl group into account.
Transcribed Image Text:
Cycloheptatrienone Cyclopentadienone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
0 60 68 D Cycloheptatrienone Cyclopentadienone A B As in the previo...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Cycloheptatrienone (I) is very stable. Cyclopentadienone (II) by contrast is quite unstable and rapidly undergoes a Diels-Alder reaction with itself. (a) Propose an explanation for the different...
-
The carbonyl group in D-galactose may be isomerized from C1 to C2 by brief treatment with dilute base (by the enediol rearrangement, Section 23-8). The product is the C4 epimer of fructose. Draw the...
-
Explain why meetings can be so inefficient. Given this, explain how effective meetings can be run.
-
Interpret the graph COVID-19 LITERACY SCALE 90 80 70 60 666 6 50 40 30 20 10 01 02 Q3 04 Q5 06 07 Q8 a9 Q10 Q11 Q12 Q13 Q14 Q15 Q16 Q17 Science Non-Science Figure 1. COVID-19 LITERARY SCALE
-
What benefits does UPS derive from using Orion to help it make drivers' work more efficient?
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
To test whether sales have been recorded, a sample should be drawn from a file of; a. Purchase orders. b. Sales orders. c. Sales invoices. d. Bills of lading. Choose the correct answer.
-
Greenock Limited has the following information available for accruals for the year ended December 31. 2017. The company adjusts its accounts annually. 1. The December utility bill for $425 was...
-
As a leader, what kind of extrinsic and intrinsic motivators could you provide for your team? How can you balance keeping your team happy if they are asking for increased compensation when you are...
-
The Metropolitan Transit Authority (MTA) has just opened a new subway line (the Orange Line) in its underground transportation network. The Orange Line had a capital investment of $20 million,...
-
Cyclopropanone is highly reactive because of its large amount of angle strain, but methylcyclopropenone, although even more strained than Cyclopropanone, is nevertheless quite stable and can even be...
-
Which would you expect to be most stable, cyclononatetraenyl radical, cation, or anion?
-
Does franchising have a bright or a dim future in the United States? Make your answer as substantive and thoughtful as possible.
-
Using tensorial symbols, establish the following vector identity: Vx (AxB) = (BV)-(AV)B+AV.B) B(V.A).
-
A planet is located at < - 3 * 1 0 1 0 , 8 *1 0 1 0 , - 7 * 1 0 1 0 > m . A star is located at < 6 * 1 0 1 0 , - 2 *1 0 1 0 , 1 * 1 0 1 0 > m . What is the vector pointing from the star to the planet?
-
in pm What is 3.7 10-11 m in units of pm? pm
-
The Wave master 4 0 0 0 T M wave machine creates harmonic water waves that propagate radially outward from the center of a cylindrical pool. Shown above are all the wave crests produced by this...
-
Show that the ODE x + xe2t sin x + 10 in R with x(0) = 0 has a unique solution. 6. PROBLEM 6 Solve the ODE, for x = x(t), x + x = ex.
-
The speeds of an electron and a proton have been measured to the same uncertainty. Which one has a larger uncertainty in position? A. The proton, because it's more massive. B. The electron, because...
-
Calculate Total Contribution Margin for the same items. Total Revenue Total Variable Costs Total Contribution Margin $50.00 a. $116.00 $329.70 b. $275.00 $14,796.00 $7,440.00 c. $40,931.25 d....
-
Give the general expression for the equilibrium constant of the following generic reaction: aA+ bB cC + dD
-
Draw the structure of the predominant form of (a) isoleucine at pH 11 (b) proline at pH 2 (c) arginine at pH 7 (d) glutamic acid at pH 7 (e) A mixture of alanine, lysine, and aspartic acid at (i) pH...
-
Write the complete structures for the following peptides. Tell whether each peptide is acidic, basic, or neutral. (a) Methionylthreonine (b) Threonylmethionine (c) Arginylaspartyllysine (d)...
-
The following structure is drawn in an unconventional manner. (a) Label the N terminus and the C terminus. (b) Label the peptide bonds. (c) Identify and label each amino acid present. (d) Give the...
-
Ida Company produces a handcrafted musical Instrument called a gamelan that Is similar to a xylophone. The gamelans are sold for $949. Selected data for the company's operations last year follow:...
-
There is a bonus with the following characteristics: nominal value $98.650.000 annual coupon rate 4.5% term 5 years, tir 6.5% A) Calculate the price of the bond assuming it is zero coupon B)...
-
A project that costs $4,450 to install will provide annual cash flows of $1,500 for each of the next 6 years. a. What is NPV if the discount rate is 11%? b. How high can the discount rate be before...

Study smarter with the SolutionInn App


