Cyclopropanone is highly reactive because of its large amount of angle strain, but methylcyclopropenone, although even more
Question:
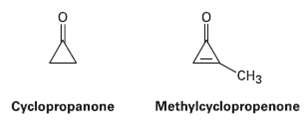
Cyclopropanone is highly reactive because of its large amount of angle strain, but methylcyclopropenone, although even more strained than Cyclopropanone, is nevertheless quite stable and can even be distilled. Explain, taking the polarity of the carbonyl group intoaccount.
Transcribed Image Text:
"CHз Cyclopropanone Methylcyclopropenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
00 A 8 0 CH3 B CH3 In resonance structure A methylcyclopropenone is a cyclic conjugated com...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The polarization of a carbonyl group can be represented by a pair of resonance structures: Cyclopropenone and cycloheptatrienone are more stable than anticipated. Cyclopentadienone, however, is...
-
Explain why, for more than a decade, a massive amount of money flowed into the United States. Compare and contrast your explanation with that of the President. Most economists agree that the problems...
-
Because a center equilibrium is stable but not asymptotically stable, nonlinear perturbation can have different outcomes. shown in Problems I 1 and 12. 1. Determine the stability of the equilibrium...
-
Great ride (GR) is in the business of manufacturing and selling high-end vehicles. GR signs a deal with the CEO of a consulting firm for a luxury SUV. You are the long-time Controller for GreatRide...
-
What challenges does the system pose for drivers and their managers?
-
(a) The hydrogen atoms attached to C3 of 1,4-pentadiene are unusually susceptible to abstraction by radicals. How can you account for this? (b) Can you provide an explanation for the fact that the...
-
Tracing copies of sales invoices to shipping documents provides evidence that: a. Shipments were recorded as receivables. b. Billed sales were shipped. c. Debits to the accounts receivable were for...
-
Root Shoe Company makes loafers. During the most recent year, Root incurred total manufacturing costs of $26,400,000. Of this amount, $2,100,000 was direct materials used and $19,800,000 was direct...
-
Determine the correct amount of net income for August and the total assets, liabilities, and owner's equity at August 31. In addition to indicating the corrected amounts, indicate the effect of each...
-
1. Why do you think the regions are not responding to Wilkess initiative for change? What did Wilkes do wrong with respect to implementing the change? 2. Should Wilkes solicit more active support...
-
Draw an energy diagram for the three molecular orbitals of the cyclopropenyl system (C3H3). How are these three molecular orbitals occupied in the cyclopropenyl anion, cation, and radical? Which of...
-
Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can?t be isolated. Explain, taking the polarity of the carbonyl group into account. Cycloheptatrienone Cyclopentadienone
-
Let X 1 ,,X n be a random sample from a Poisson() distribution. a. Find the likelihood equation, L(x 1 ,,x n ;), using as the PMF. b. Find the log likelihood function and use that to obtain the MLE...
-
Following are the financial data for Binari Jaya Sdn Bhd as at 3 1 December 2 0 2 2 . Revenue (Sales) RM 1, 370, 100 Account Receivable RM 540, 000 Long Term Loan RM 690, 000 Cost of goods sold RM...
-
1. Who are the people affected by rsum truth decisions? 2. Why might a job seeker have a duty to blur parts of his or her work history? 3. Why might an egoist lie on the rsum, and why not? 4. Does an...
-
Find the following. 2. 3 y(t) + 3y(t) 3+ = x(t) + 3 x(t) 3. y(t) - 1/2 y(t) x(t) = dt d 4. dt d 5. dt y(t) + 1/2 y(t) + y(t) y(t) + 1/2 y(t) = x(t) + x(t) = x(t)
-
A 2-meter-tall basketball player attempts a goal 12.4 meters from the basket that is 3.05 meters high. If he shoots the ball at a 55 angle, at what initial speed must he throw the basketball so that...
-
How is an x-y "graph for the eye" different from x vs. t or y vs. t graphs?
-
A gold cathode is illuminated with light of wavelength \(250 \mathrm{~nm}\). It is found that the current is zero when \(\Delta V=1.0 \mathrm{~V}\). Would the current change if a. The light intensity...
-
Will the prediction interval always be wider than the estimation interval for the same value of the independent variable? Briefly explain.
-
Nitrogen monoxide, a pollutant in automobile exhaust, is oxidized to nitrogen dioxide in the atmosphere according to the equation: Find K c for this reaction. 2 NO(g) + 0(8) 2 NO(8) Kp 2.2 x 102 at...
-
Suggest a method for the synthesis of the unnatural D enantiomer of alanine from the readily available L enantiomer of lactic acid. CH,-CHOH-COOH lactic acid
-
Show how you would use the Gabriel-malonic ester synthesis to make histidine. What stereochemistry would you expect in your synthetic product?
-
Show how you would use the Strecker synthesis to make tryptophan. What stereochemistry would you expect in your synthetic product?
-
The market is expected to return 15 percent next year and the risk-free rate is 7 percent. What is the expected rate of return on a stock with a beta of 1.3? The covariance of the market's returns...
-
A stock's current price is 145.05. A put option with an exercise price of 120 and maturity of 3 months is currently priced at $ 28.83. What is the option's time value?
-
An investor put 60 percent of his money into a risky asset offering a 10 percent return with a standard deviation of return of 8 percent, and he put the balance of his risk-free asset offering 5...

Study smarter with the SolutionInn App


