Dichiorocarbene can be generated by heating sodium trichioroacetate. Propose a mechanism for the reaction, and use curved
Question:
Dichiorocarbene can be generated by heating sodium trichioroacetate. Propose a mechanism for the reaction, and use curved arrows to indicate the movement of electrons in each step. What relationship does your mechanism bear to the base-induced elimination of HC1 fromchloroform?
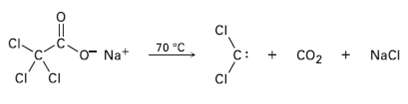
Transcribed Image Text:
CI 70 "C Na+ NaCI C: CO2 ci CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (17 reviews)
444 C 2 C In step 1 carbon dioxide is lost from the trichloroace...View the full answer

Answered By

Diksha Bhasin
I have been taking online teaching classes from past 5 years, i.e.2013-2019 for students from classes 1st-10th. I also take online and home tuitions for classes 11th and 12th for subjects – Business Studies and Economics from past 3 years, i.e. from 2016-2019. I am eligible for tutoring Commerce graduates and post graduates. I am a responsible for staying in contact with my students and maintaining a high passing rate.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Propose a mechanism for the reaction of (a) 1-methylcyclohexanol with HBr to form 1-bromo-1-methylcyclohexane. (b) 2-cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane?
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
Hydrocarbon A has the formula C 9 H 12 and absorbs 3 equivalents of H 2 to yield R, C 9 H 18 , when hydrogenated over a Pd/C catalyst. On treatment of A with aqueous H 2 SO 4 in the presence of...
-
Below are five questions from a survey of MBA students. Answers were written in the blank at the left of each question. For each question, state the data type (categorical, discrete numerical, or...
-
An electrical contact material is produced by infiltrating copper into a porous tungsten carbide (WC) compact. The density of the final composite is 12.3 g/cm3. Assuming that all of the pores are...
-
Draw an ogive for the frequency distribution in Example 2. Data from Example 2 Using the frequency distribution constructed in Example 1, find the midpoint, relative frequency, and cumulative...
-
A gear reduction unit uses the countershaft shown in the figure. Gear A receives power from another gear with the transmitted force FA applied at the 20_ pressure angle as shown. The power is...
-
A concave spherical mirror has a focal length of 6.00 cm. If an object is placed at a distance of 24.0 cm from the mirror, calculate the image distance and characteristics of the image including...
-
In the ZZZZ Best case, explain the kinds of evidence that the auditors gathered and how the auditors failed. just write one or two paragraph to support ZZZZ Best Case On May 19, 1987, a short article...
-
How would you distinguish between the following pairs of compounds using simple chemical tests? Tell what you would do and what you would see. (a) Cyclopentane and cyclopentane (b) 2-Hexene and...
-
(a) How many degrees of unsaturation does a-terpinene have? (b) How many double bonds and how many rings does it have? (c) Propose a structure fora-terpinene. CCH-CH2CCHCH3 C-C H ...
-
Write an equation of each line that satisfies the given conditions. Give the equation (a) In slope-intercept form (b) In standard form Through (0,0) and (1,4)
-
A 100-Year Bond does not have appreciably more price risk than a 30-Year Bond. Why is this?
-
Long-term employees may find themselves motivated by loyalty to their superiors rather than the mission of the organization. This phenomenon seems to be more likely in criminal justice organizations...
-
Describe characteristics of addictive populations. Discuss the challenges of working with a specific addictive population such as alcoholism or drugs. Discuss the theoretical perspective(s) that you...
-
Please identify 2 pros (advantages) and 2 cons (disadvantages) of investing in real estate as a Private Equity Investor within a syndicate or consortium of equity investors (that is, you are an...
-
Your reflection should be four to six paragraphs in length and may consider some or all of the following questions: What message or statement do you believe director Spike Lee was trying to make with...
-
Consider a swap when fixed is paid and floating is received. If the term structure of interest rates is upward sloping, do the early exchanges have a positive or negative value?
-
A company produces earbuds. The revenue from the sale of x units of these earbuds is R = 8x. The cost to produce x units of earbuds is C = 3x + 1500. In what interval will the company at least break...
-
How does each of the three major bonding theories (the Lewis model, valence bond theory, and MO theory) define a single chemical bond? A double bond? A triple bond? How are these definitions similar?...
-
Consider the following well-known reaction of glycols (vicinal diols). (a) In this reaction, what species is oxidized? What species is reduced? Explain how you know. (b) How many electrons are...
-
CompolnA A, C7H14, decolorizes Br, in CH2CI2? and reacts with BH3, in THF followed by H'O2/OH- to yield compound B. When treated with KMnO4, then H3O+, B is oxidized to a carboxylic acid C that can...
-
CompolnA A, C7H14, decolorizes Br, in CH2CI2? and reacts with BH3, in THF followed by H'O2/OH- to yield compound B. When treated with KMnO4, then H3O+, B is oxidized to a carboxylic acid C that can...
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
Marcia Stanton is the new manager of the materials storeroom for Taylor Manufacturing. Marcia has been asked to estimate future monthly purchase costs for part #696, used in two of Taylor's products....
-
Miller Company's contribution format income statement for the most recent month is shown below: Sales (37,000 units) Variable expenses Contribution margin Fixed expenses Net operating income...

Study smarter with the SolutionInn App


