Draw all of the stereo isomers of these compounds: OH a) CHCH=CHCHCH3 b) CHCH=CHCHCH=CHCH
Question:
Draw all of the stereo isomers of these compounds:
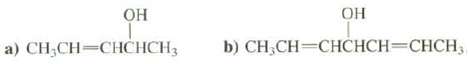
Transcribed Image Text:
OH a) CH₂CH=CHCHCH3 ОН b) CH₂CH=CHCHCH=CHCH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 36% (11 reviews)
a HC H b H3C ...View the full answer

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw all of the stereo isomers for 2, 3-dichlorobutane. Indicate which rotate plane-polarized light and which are meso.
-
Draw all of the stereo isomers of 1, 2-dimethylcyclopropane. Explain which rotate plane-polarized light.
-
All of the stereo isomers of 1, 2, 3, 4, 5, 6-hexachlorocyclohexane have very similar rates of E2 reaction except the following stereo isomer, which reacts about 7000 times more slowly than the...
-
int Temp [10] = { 22, 30, 40,28,20,60}; For the above code, answer the following questions i) Find the size of Temp array. ii) How many memory locations are present in Temp array ? iii) Find the...
-
The Stately Retail Group owns the EzyShop chain of discount department stores. The EzyShop store in Bondi Junction consists of several departments. The revenues and costs for each department are...
-
Sonata Manufacturing Corporation decided to expand its operations and open a new facility in Illinois. Rather than constructing a new plant, Sonata negotiated a contract to purchase an existing...
-
What is a System Operations Dictionary?
-
Rolling Hills Golf Inc. was organized on July 1, 2012. Quarterly financial statements are prepared. The trial balance and adjusted trial balance on September 30 are shown here. Instructions(a)...
-
Prepare a detailed explanation of use of views in database security as well as database system administration. Explain the ways views can be implemented, why views would be implemented. Include at...
-
2. In the following circuit, there exist a constant and uniform magnetic field in the shaded region. A switch-key can periodically switch between 1 and 2 with time period T. When switch key touches...
-
Determine the number of stereo isomers for these compounds: a) OH OH b) d) CHCH3 CHCH
-
Explain whether or not these compounds would rotate plane-polarized light: ) ) i) OH H C. H3C H.C. HC H HO OH CH3 -OH - CH3 CH3 CH3 CH CH3 b) f) h) HC d) Cl-C-CCI H C "OH H H CH H CHO CH CH3 OH...
-
The Mach number _______. (Select all that are correct). a. Is the ratio V/c, where c = specific heat b. Is the ratio V/c, where c = the speed of sound c. Depends on the velocity, V, of the fluid...
-
An ice-cream company has a new business idea: specially equipped ice-cream trucks can deliver the company's ice-creams to customers' homes and create custom ice-cream flavors right there. Seems like...
-
The Balance Sheet for the current year of a company is given below (All the figures are in Lacy- Liabilities Assets Equity Capital Retained EarningsP Term Loan 100 Land & Building 120 Machinery 160...
-
3. Calculate the total particulate mass concentration (g/m3) of the diesel engine exhaust aerosol when the emissions were found by measuring two particle modes (you can assume the particles are...
-
Q4 (a) (b) (c) A mechanism for a trolley system from a manufacturing process is illustrated in Figure Q4. The trolley has a mass 5m, a displacement x, and is restrained by a spring of stiffness k...
-
A pipe handles a flow rate of 35 gpm. Find the minimum inside diameter that will provide an average fluid velocity not the exceed 20 ft/s. Problem 2 A steel tubing has a 1.5-in outside diameter and a...
-
What is the master budget, and why is it useful?
-
Frontland Advertising creates, plans, and handles advertising campaigns in a three-state area. Recently, Frontland had to replace an inexperienced office worker in charge of bookkeeping because of...
-
In Exercises 16, compute '(x) using the limit definition. (x) = 1 x 1
-
A Assign R or S configuration to each chirality center in pseudoephedrine, an over-the-counter decongestant found in cold remedies (blue =N).
-
Which of the following compounds are chiral? Draw them, and label the chirality centers. (a) 2, 4-Dimethylheptane (b) 5-Ethyl-3, 3-dimethylheptane (c) cis-l, 4-Dichlorocyclohexane (d) 4,...
-
A Draw chiral molecule that meet the following descriptions: (a) A chloro alkane, C5H11C1 (b) An alcohol, C6H14O (c) An alkene, C6H12 (d) An alkane, C8H18
-
If the nominal interest rate is 14%, and inflation is 4%, what is the real interest rate? 22.A bond investment yielded 8%. If inflation was 3%, what real return did the bond offer? 23.The following...
-
Bierce Corporation has two manufacturing departments--Machining and Finishing. The company used the following data at the beginning of the year to calculate predetermined overhead rates: Estimated...
-
Discuss how communication channels have changed over the years due to technology.

Study smarter with the SolutionInn App


