Draw the important resonance structures for these species and discuss the contribution of each to the resonance
Question:
Draw the important resonance structures for these species and discuss the contribution of each to the resonance hybrid. Explain whether the species has a large or a small amount of resonance stabilization?
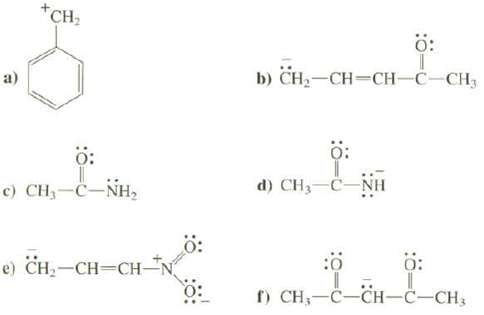
Transcribed Image Text:
a) +CH₂ Ö: c) CH,—C−NH, e) CH₂-CH=CH- b) CH₂-CH=CH-C-CH3 Ö: d) CH₂-C-NH Ö: :0 Ö: f) CH-C-CH-C-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a All of these resonance structures are important Although the carbocation is unstable because it do...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. Discuss the relative...
-
For each of these ions, draw the important resonance forms and predict which resonance form is likely to be the major contributor. (a) (b) (c) CH2 CH CH2 HN
-
Show the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. ) d) . CH3 H- b) CH3-N + H -H...
-
Taxpayer sold the following capital assets during the year: Asset # 1: $60,000 long-term capital gain Asset # 2: $10,000 long-term capital loss Asset # 3: $50,000 short-term capital gain Asset # 4:...
-
Explain how the terms controllability and attributability are relevant when evaluating the performance of business unit managers and business units?
-
Roedel Electronics produces tablet computer accessories, including integrated keyboard tablet stands that connect a keyboard to a tablet device and holds the device at a preferred angle for easy...
-
What is an SRR and what is it intended to accomplish?
-
The following information, taken from the books of Herman Brothers Manufacturing represents the operations for January: The job cost system is used, and the February cost sheet for Job M45 shows the...
-
Suppose 1 needed to graph the equation 30a60b120 with a on the vertical axis and b on the horizontal axis. The vertical intercept of the line would be...?
-
In Shady Company, materials are entered at the beginning of each process. Work in process inventories, with the percentage of work done on conversion costs, and production data for its Sterilizing...
-
In these examples the additional structure or structures are not important contributors to the resonance hybrid for the compound represented by the first structure, explain. a) 8-8 c) CH-C=N: b) :0...
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
In Exercises 1 through 26, use integration by parts to find the given integral. (t-1)e- dt
-
a) Why is inventory management important? How does mismanaged inventory leads to costs to any organisation? (10 marks) Give examples to justify your answers. (Please answer in 200-300 words). b) Reem...
-
A table has sold for $670 (15% VAT inclusive). Calculate the new setting price of this table if the VAT was raised to 17.5%
-
Consider the labor market with the following demand and supply, respectively: Qd Qs 2 100+ N P = 2P Where: P represents hourly wage (dollars). In your graph, use the natural slopes for the Demand and...
-
You deposit $480 each week into an account earning 3.7% interest compounded weekly. Use the TVM Solver on the TI calculator. Round to the nearest cent (two decimal places). a) How much will have...
-
Given the following is a probability distribution for returns on a security A, and a proxy for the market, M: State 1 2 3 4 Probability 10% 40% 40% 10% RA 12% 6% 1% -2% RM 15% 8% 4% -6% a. Calculate...
-
Imagine that you are an independent musician that plays the drums. You are on tour with a band, but it is your responsibility to get from place to place with your gear. For short trips, you need a...
-
Air pollution generated by a steel mill is an example of a) a positive production externality. b) a negative production externality. c) a public good. d) the free-rider problem. State and local taxes...
-
For what values of p is the series convergent? n=1 1 nP
-
Malic acid, C4H6O5, has been isolated from apples. Because this compound reacts with 2 molar equivalents of base, it is a dicarboxylic acid. (a) Draw at least five possible structures. (b) If malic...
-
Formaldehyde, H2C = O, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane, C3H6O3, which, surprisingly enough, has no...
-
Increased substitution around a bond leads to increased strain. Take the four substituted butanes listed below, for example. For each compound, sight along the C2-C3 bond and draw Newman projections...
-
Graph the inequality. x + y -4
-
Find the slope of the line through the points (-3, 7) and (-9,2). Enter your answer as a simplified improper fraction, if necessary. Do not include "m="in your answer.
-
What is the maximum number of linearly independent vectors that can be found in the nullspace of: A = 120 3 1 24-15 4 36-1 85 4 8 1 12 8

Study smarter with the SolutionInn App


