Draw the important resonance structures for these species. Use the curved arrow convention to show how the
Question:
Draw the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. Discuss the relative contribution of each to the resonance hybrid and the overall resonance stabilization of the species.
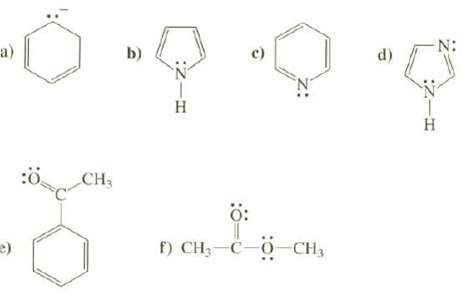
Transcribed Image Text:
a) e) :Ö CH₂ b) :ZIH H c) ő: f) CH₂-C-O-CH₂ d) :Z-H Η N:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
a Each of these structures makes a similar contribution to the resonance hybrid because they have si...View the full answer

Answered By

Dominic Joseph
Education:
Bachelors' in Mechanical Engineering (2012-2016)
College - College of Engineering Trivandrum, Kerala, India
CGPA - 8.03/10
Graduated with first class distinction in 2016
Tutoring experience:
Expert Tutor (2019 - Present)
I have significant experience in tutoring multiple high school / college / post graduate students and working professionals in several fields, through multiple tutoring platforms
Subjects Taught:
• Algebra, Number systems and Complex Numbers
• Linear Algebra
• Calculus and Multivariable calculus
• Differential Equations (ODE and PDE)
• Statistics and Probability
• Data Science
• R programming, Python Programming, SAS programming
Tutored several Highschool/College students offline in the field of mathematics and statistics
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the important resonance structures for these species and discuss the contribution of each to the resonance hybrid. Explain whether the species has a large or a small amount of resonance...
-
Show the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. ) d) . CH3 H- b) CH3-N + H -H...
-
Show the important resonance structures for these compounds. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. N: d) CH-C b) 0-H O-H c)...
-
Necked Amber purchased a bond for $1,038.90 exactly two years ago. At that time, the bond had a maturity of five years and a coupon rate of 10% (paid semi-annually). Assuming the rates below are the...
-
Indicate the type of responsibility centre that is most appropriate for each of the following: 1. A cinema in a company that operates a chain of theatres. 2. A television station owned by a large...
-
A grocery store introducing items from Italy is interested in analyzing buying trends of new international items: prosciutto, pepperoni, risotto, and gelato. a. Using a minimum support of 100...
-
Using the data in the file, test the null hypothesis that urban and rural residents have the same attitudes toward gun control. Use the T.TEST function to test the hypothesis. Assume that the...
-
The L. Young & Sons Manufacturing Company produces two products, which have the following profit and resource requirement characteristics: Last months production schedule used 350 hours of labor in...
-
You have a Microsoft SQL Server instance that runs in an on-premises datacenter. You plan to deploy a SQL Server instance to an Azure virtual machine and implement an Always On availability group...
-
Determine the mass moment of inertia of the steel machine element shown with respect to the z axis. (The density of steel is 7850 kg/m 3 .) 360 mim S0 mm 200 mm 240 mm 50 mm 100 mm
-
Discuss the actual structure and the amount of resonance stabilization for the examples shown in problem 3.14.
-
Show energy level diagram for the MOs of these compounds? a) H-C=N: b) H 0: C CH3 ) CHNH,
-
What are the major differences between quantitative and qualitative research?
-
2. Two firms- -an incumbent I and an entrant E- can produce an identical good at a cost of c> 0 per unit. The incumbent chooses its quantity q1 0 first. The entrant chooses its quantity q 0 after...
-
QUESTION 2 (25 points) Consider a city with a total of n = 2500 commuters who travel to work everyday either using a freeway or their preferred alternate mode of travel. The associated graph depicts...
-
Suppose the monopolist can price discriminate after the 4th unit. That is, the first four units are sold at a price of $27. If they want to sell a 5th unit, they charge $27 from the first four units,...
-
Please complete the rest of the operating budget analysis for CHEWY INC (CHWY via New York Stock Exchange) using your own scholarly source. I included the excel link to the financial statements from...
-
FIN 3010 ASSIGNMENT 1 QUESTION ONE (i) "Discounted payback ensures that you don't accept an investment with a negative NPV but it can't stop you from rejecting projects with a positive NPV"...
-
Apple. Apple seeks energetic, dedicated students with a passion to educate others about Apple products. The company prefers students who excel at time management, have a marketing or sales...
-
Use the following data to answer the next two (2) questions: Product 1 Product 2 Product 3 Direct Material Cost $25,000 $30,000 $35,000 Direct Labor Cost $30,000 $40,000 $50,000 Direct Labor Hours...
-
Find a power series representation of x 3 /(x + 2).
-
Draw structures that meet the following description (there are many possibilities): (a) Three isomers with the formula C8H18 (b) Three isomers with the formula C4H8O2
-
Draw structures of the nine isomers of C7H16.
-
In each of the following sets, which structures represent the same compounds and which represent different compounds? CH Br C (a) " , n CHH CH3CHCHCH3 Br Br CH . (b) , " (c) CH CH2CH3 CH H,H,, ,...
-
Wal-Mart's frustrations in India Seven years ago Wal-Mart set out to be India's top retailer by 2015. The business plan was called Project Jai Ho, a Hindi phrase meaning "let there be victory". But...
-
a) identify potential mitigating strategies the organization could employ to reduce the impact of their highest ranked social risks. (2 marks) Risk The effect on the community and environment due to...
-
What is a good guideline for the amount of information to include in a message?

Study smarter with the SolutionInn App


