Draw the most stable chair conformation ofdihydrocarvone CH3 -H Dihydrocarvone
Question:
Draw the most stable chair conformation ofdihydrocarvone
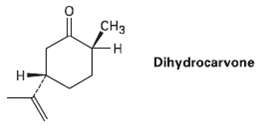
Transcribed Image Text:
CH3 -H Dihydrocarvone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 52% (17 reviews)
CH3 H Dihydrocarvone I ...View the full answer

Answered By

Surojit Das
I have vast knowledge in the field of Mathematics, Business Management and Marketing. Besides, I have been teaching on the topics Management leadership, Business Administration, Human Resource Management, Business Communication, Accounting, Auditing, Organizer Behaviours, Business Writing, Essay Writing, Copy Writing, Blog Writing since 2020. It is my personality to act quickly in any emergency situations when students need my services. I am very professional and serious in every questions students asked me at the time of dealing any projects. I have been serving detailed, quality, properly analysed research paper through the years.
4.80+
91+ Reviews
278+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the most stable chair conformation of -D-galactose.
-
Draw the most stable chair conformation of menthol, and label each substituent as axial orequatorial. Menthol (from peppermint oil) - CH
-
Draw the most stable chair conformation of 1-bromo-2-chlorocyclohexane, if the coupling constant between hydrogens on C1 and C2 was found to be 7.8 Hz (J1,2 = 7.8 Hz).
-
1. Create your Dream Board (all the things you want to buy or establish in the future). Specify the amount of each item. 2. Search the salary of your prospective careers 5 years from now. 3. With the...
-
What is a top management team, and how does it affect a firm's performance and its abilities to innovate and design and implement effective strategic changes?
-
For the network of Fig. 4.125, determine: a. IB. b. IC. c. VCE. d. VC. 9+16 V 12 k VE B-120 Vca 9.1 k 15 k 6-12 V
-
The following data give the means and ranges of 25 samples, each consisting of 4 compression test results on steel forgings, in thousands of pounds per square inch: (a) Use these data to find the...
-
Del Monte has a long and rich tradition in the American food processing industry. It is perhaps best known for packaging canned fruits and vegetables. Part of its success has involved acquiring other...
-
Gonzalo works in the budget department of a company that manufactures and markets women's shoes. He is responsible for preparing the company's master budget, however, Adn, the director of the...
-
Last year, at Northern Manufacturing Company, 200 people had colds during the year. One hundred fifty-five people who did no exercising had colds, and the remainders of the people with colds were...
-
Suggest a mechanism by which ?-ionone is transformed into ?-ionone on treatment with acid. H3o -Ionone B-Ionone
-
As a general rule, equatorial alcohols are esterified more readily than axial alcohols. What product would you expect to obtain from reaction of the following two compounds with 1 equivalent of...
-
Juan and Celina are married, file a joint return, and have AGI of $148,000. On April 1, 2010 they purchased their first home for $118,000 and claimed the homebuyer credit on their 2010 tax return. a....
-
Draw the logic gate symbol for a 4-input AND gate. Write the Boolean function and truth table. [20] Deduce the Right-hand side of three variable DeMorgan's theorem shown below using Boolean axioms...
-
Galaxy Inc. reported net income for accounting purposes of $1,130,000. In calculating this amount, the following items were deducted: 1- Income tax expense 2- Interest expense (Includes $7,000 of...
-
1. Find the equation of motion using any of the following methods; a. Using Newton's Method, b. Using the D'Alembert method c. Using the Energy Method d. Using Lagrange's Method, II. Find the natural...
-
You have been asked to present the valuation consequences of this project to the Board of Directors. The Board is particularly interested in viewing this project from the perspective of the...
-
(60pts) This class has static methods that will help us evaluate Lisp arithmetic expressions. Before a List expression is evaluated, we have to make sure that it is a valid expression. The following...
-
Let \(\left(\mathscr{F}_{t} ight)_{t \geqslant 0}\) be an admissible filtration for the Brownian motion \(\left(B_{t} ight)_{t \geqslant 0}\). Mimic the proof of Lemma 2.14 and show that for each...
-
In Exercises 1558, find each product. (9 - 5x) 2
-
Redo Problem 3.32 using Aspen Plus. Problem 3.32 Nitrogen can be liquefied using a Joule-Thomson expansion process. This is done by rapidly and adiabatically expanding cold nitrogen gas from high...
-
Divide the structures of (a) myrcene and (b) b-pinene (page 453) into their component isoprene units.
-
Notice (from your answer to Problem 15.8) that all of the isoprene units in farnesol and retinal are joined in a head-to-tail manner, but that they are not all arranged that way in squalene and...
-
Ball-and-stick structures of the odiferous components of (a) spearmint and (b) caraway seed are shown below. What are the stereochemical relationships between these compounds? () (a)
-
Give an example of one piece of financial information that may be used differently for two stakeholders (users) and explain how it would be used and presented differently?
-
Complete common -size statements (balance sheets) using the data below on page 246. You will be required to create a spreadsheet in MS Excel to display the statements (balance sheets).? In a 1 to 2...
-
A sail boat in the Great South Bay travels 2 miles east and then 4 miles south to get to Fire Island. What is its distance and displacement?

Study smarter with the SolutionInn App


