Draw the p orbital's that compose the conjugated part of these molecules: a) :-CH3 b) CH,=CHNH, c)
Question:
Draw the p orbital's that compose the conjugated part of these molecules:
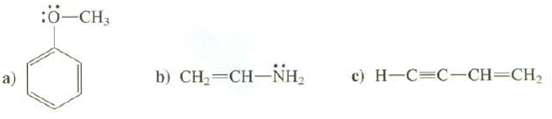
Transcribed Image Text:
a) :Ö-CH3 b) CH,=CH–NH, c) H-C-C-CH=CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a CH3 b H 181 H c H fir 8th CH...View the full answer

Answered By

Muhammad Khurram
I have strong General Management skills to apply in your projects. Over last 3 years, I have acquired great knowledge of Accounting, Auditing, Microsoft Excel, Microsoft PowerPoint, Finance, Microsoft Project, Taxation, Strategic Management, Human Resource, Financial Planning, Business Planning, Microsoft Word, International Business, Entrepreneurship, General Management, Business Mathematics, Advertising, Marketing, Supply Chain, and E-commerce. I can guarantee professional services with accuracy.
4.80+
249+ Reviews
407+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw an acceptable Lewis electron dot diagram for these molecules that violate the octet rule. a. NO2 b. XeF4
-
Draw an acceptable Lewis electron dot diagram for these molecules that violate the octet rule. a. BCl3 b. ClO2
-
Draw an acceptable Lewis electron dot diagram for these molecules that violate the octet rule. a. POF3 b. ClF3
-
V = 6V 1052 3F 1-what value will be the voltage across the capacitor after 2 times constant? 2-what value will be the voltage across the capacitor after 6 seconds? 3-when will the capacitor be fully...
-
Explain the benefits of using transfer pricing within organizations?
-
Tunningley Services is establishing a new business to serve customers in the Ohio, Kentucky, and Indiana region around the Cincinnati Ohio area. The company has identified 15 key market areas and...
-
Repeat Problem 10.27 if the Froude number is 2.75. Problem 10.27 Water flows in the river shown in Fig. P10.27 with a uniform bottom slope. The total head at each section is measured by using Pitot...
-
You work at a firm on Wall Street that specializes in mergers, and you are the team leader in charge of getting approval for a merger between two major beer manufacturers in the United States. While...
-
Evaluate the limit: lim h-0 r(t + h) -r(t) h r'(t)=000 for r(t)=(t2, sint, -9)
-
Betty Cardinal runs Cardinals Book Shop in a downtown location. As her newly hired accountant, your task is to do the following (substituting HST at 13% for GST at 5% if your instructor so directs):...
-
What is the hybridization at all atoms, except hydrogen's, in these compounds? a) H H H H H H H H CH 4.6 c) HC-N-CH3 : : g) CHC-OH h) CHC-NHCH,
-
Show the important resonance structures for these compounds. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. N: d) CH-C b) 0-H O-H c)...
-
A single-server waiting system is subject to a homogeneous Poisson input with intensity \(\lambda=30\left[h^{-1} ight]\). If there are not more than 3 customers in the system, the service times have...
-
A particle carrying charge \(+10.00 \mathrm{nC}\) sets the origin of a rectangular coordinate system. Taking the electrostatic potential to be zero at infinity, locate the distance from the origin of...
-
What is bivariate regression?
-
Given that $90 %$ of criminal defendants are indigent, do you think more of an effort should be made to increase the money that is paid to counsel appointed to represent these people to ensure that...
-
Consider a system described by the quasispin model of Problem 31.3 for $G>0$ with two identical fermions in a single $j$-shell. Show that the allowed seniorities are $v=0,2$, the allowed angular...
-
A point charge \(q=+5 \mathrm{nC}\) is at the center of a thick conducting spherical shell of internal radius \(10 \mathrm{~cm}\) and external radius \(12 \mathrm{~cm}\). (a) What is the magnitude...
-
(a) Test whether \(\mu_{1}
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
For what values of x does the series converge? 00 3 n=1 (x 3)" n
-
Explain why each of the following names is incorrect: (a) 2, 2-Dirnethyl-6-ethytheptane (b) 4-Ethyl-5, 5-dirnethylpentane (c) 3.Ethyl-4, 4-dimcthylhexane (d) 5, 5, 6-Trimcthyloctane (e)...
-
Propose structures and give IUPAC names for the following: (a) A diethyldimethyihexane (b) A (3-methylbutyl)-substituted alkane
-
Consider 2-methylbutane (isopentane) Sighting along the C2C3 bond: (a) Draw a Newman projection of the most stable conformation. (b) Draw a Newman projection of the least stable conformation. (c)...
-
The Power of Trade and Comparative Advantage: Work It Out 3 ? Here's another specialization and exchange problem. This problem is wholly made-up, ? so that you won't be able to use your intuition...
-
Government spending as a fiscal policy tool is used to: A) ?Decrease the national debt B) ?Directly stimulate economic activity by increasing demand C) ?Reduce inflation D) ?Lower interest rates
-
Automobile demand has fallen during a recent recession, and Ana has been laid off from her job on the assembly line. Rajiv recently lost his job as a waiter at a local restaurant. A recent increase...

Study smarter with the SolutionInn App


