Show the important resonance structures for these compounds. Use the curved arrow convention to show how the
Question:
Show the important resonance structures for these compounds. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure.
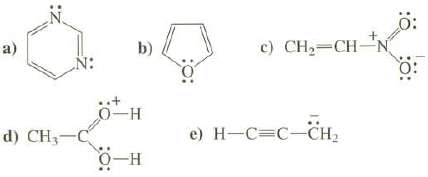
Transcribed Image Text:
N: d) CH₂-C b) 0-H O-H c) CH₂=CH-N e) H-C=C-CH₂ Ö: 0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
a The unshared electron pairs on the nitrogens are not part of the conjugated pi system b One uns...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. ) d) . CH3 H- b) CH3-N + H -H...
-
Draw the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. Discuss the relative...
-
Draw the important resonance structures for these species and discuss the contribution of each to the resonance hybrid. Explain whether the species has a large or a small amount of resonance...
-
You are developing an industrial building with a gross building area of 150,000 sf. The building efficiency ratio is 75%. The market gross rent is $25 psf. The vacancy rate is 5%; the cap rate is 5%;...
-
How might real-time reporting contribute to improving the competitive advantage of an organization?
-
The PCAOB allows the auditor to use the work of others, such as internal auditors or other company personnel, to alter the nature, timing, or extent of the auditor's own testing of internal controls....
-
Explain the difference between a. Actors and stakeholders. b. Primary and supporting actors.
-
Solvo produces three types of trucks on one assembly line: the long-haul (L), the short-haul (S) and the vocational truck (V). Next month the company has orders for 500 long-haul trucks, 750...
-
Rank the following three single taxpayers in order of the magnitude of taxable income. Assume none of the taxpayers contributed to charity this year. Note: First mean highest taxable income, third...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Draw the p orbital's that compose the conjugated part of these molecules: a) :-CH3 b) CH,=CHNH, c) H-C-C-CH=CH
-
In these examples the additional structure or structures are not important contributors to the resonance hybrid for the compound represented by the first structure, explain. a) 8-8 c) CH-C=N: b) :0...
-
Find the function represented by the given power series. x2a+1 E(-1)" 22n+ (2n + 1)! n=0
-
A closed box contains 20 particles, 10 on each side of a movable partition. The particles in the left compartment are small and occupy a volume \(\delta V_{\text {left. }}\). Those in the right...
-
A box that is \(1.00 \mathrm{~m}\) long measured from left to right has a sliding partition that separates it into two compartments. The left compartment contains 10 particles, the right one contains...
-
A change in the volume occupied by a set of distinguishable particles increases the number of basic states by a factor of 6633 and increases the entropy per particle by a factor of 1. 10. How many...
-
Rank the following systems according to their entropy, lowest first: A. 1,000,000 different colors that can be assigned to each of the \(70 \times 10^{21}\) stars in the observable universe B. The...
-
The rate of change of entropy with respect to thermal energy is \(1.81 \times 10^{20}\) in a system at \(400 \mathrm{~K}\). At what absolute temperature is the rate of change twice this value?
-
The Wall Street Journal investigated online pricing and discovered that your price may vary indeed. \({ }^{41}\) Using your zip code, online retailers determine the price of your stapler, your saw,...
-
The following information is for Montreal Gloves Inc. for the year 2020: Manufacturing costs Number of gloves manufactured Beginning inventory $ 3,016,700 311,000 pairs 0 pairs Sales in 2020 were...
-
Indicate which tests should be used. n + 1 =1 33 + 4n? + 2
-
What are the relative energies of the three possible staggered conformations around the C2C3 bond in 2, 3-dimethylbutane? (See Problem 3.42)
-
Construct a qualitative potential-energy diagram for rotation about the CC bond of 1, 2-dibromoethane. Which conformation would you expect to be more stable? Label the anti and gauche conformations...
-
Which conformation of 1, 2-dibrornoethane (Problem 3.44) would you expect to have the larger dipole moment? The observed dipole moment of 1, 2-dibromoethane is = 1.0 D. What does this tell you about...
-
Company name is Walmart, Inc. here is the link https://www.sec.gov/ix?doc=/Archives/edgar/data/104169/000010416923000020/wmt-20230131.htm Overview 1. The financial statements for your company are...
-
Congress would like to increase tax revenues by 19 percent. Assume that the average taxpayer in the United States earns $54,000 and pays an average tax rate of 15 percent. Required: a. If the income...
-
Best Solutions is a retail merchandiser selling computer equipment. Best uses the gross method of accounting for inventory purchases and sales, a perpetual inventory system with LIFO inventory...

Study smarter with the SolutionInn App


