Draw the three-dimensional furanose form of ascorbic acid (Problem 25.32) , and assign R or S stereochemistry
Question:
Draw the three-dimensional furanose form of ascorbic acid (Problem 25.32), and assign R or S stereochemistry to each chirality center.
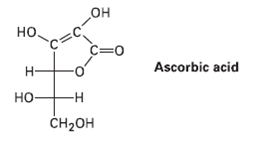
но но. c=0 Ascorbic acid н -н но CH2он
Step by Step Answer:
Ascorbic acid has an L configurat...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Organic Chemistry questions
-
Name and assign R or S stereochemistry to the product(s) you would obtain by reaction of the following substance with ethyl magnesium bromide. Is the product chiral? Is it optically active? Explain.
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
Assign R or S configuration to each chirality center in the following biologicalmolecules: (a) (b) .C 'N' N-N - CH2CH2CH2CH2CO2 Prostaglandin E, Biotin
-
a. Calculate the quick ratio of company B in FY2020. State what factors need to be considered when evaluating company B?s short-term liquidity and analyse the impact of these factors on the...
-
Read the step by- step hints and examples for writing a funding proposal at www.learnerassociates.net/proposal. Review the entire sample proposal online. What details did the writer decide to include...
-
A copper wire is stretched with a stress of 70 MPa (10,000 psi) at 20C (68F). If the length is held constant, to what temperature must the wire be heated to reduce the stress to 35 MPa (5000 psi)?
-
Under the provisions of the Companies Act 1985 an auditor's report must be attached to a company's financial statements. Is this true for all companies? Explain.
-
Alternative methods of joint-cost allocation, product-mix decisions. The Sunshine Oil Company buys crude vegetable oil. Refining this oil results in four products at the splitoft point; A, B, C, and...
-
Discuss at least two reasons why job analysis is often described as the foundation of human resources management. Provide specific examples to support.
-
Forecasting with the Parsimonious Method and Estimating Share Value Using the ROPI Model Following are income statements and balance sheets for Cisco Systems. Cisco Systems Consolidated Statements of...
-
Does ascorbic acid (vitamin C) have a D or Lconfiguration? HO . C=0 Ascorbic acid . -H - CH2OH
-
Assign R or S configuration to each chirality center in the followingmolecules: (c) (a) (b) NH2 - H3C- Br - Br - - - CH - - - -
-
StarLight Ltd. is authorized to issue 10,000, $4 noncumulative preferred shares. On January 13, it issued 3,000 preferred shares for $90 cash per share. (a) Prepare a journal entry to record the...
-
An experienced tinter can tint a car in 4 hours. A beginning tinter needs 9 hours to complete the same job. Find how long it takes for the two to do the job together.
-
X is all-equity with 300,000 shares outstanding. Current market price of one stock is $5.4. X plans to issues 500 new perpetuity bonds, face value $1000, interest rate 5%. The proceeds from the...
-
Consider a rigid structure in the sketch. The structure is clamped at point A while a 100 newton force F works on point D in DC direction. Calculate magnitude of bending moment and torsion of the...
-
For centuries women have fought the right to have a voice in a male-dominated world. Much has been said about how women should treat their bodies in recent years. In 2000 the very first uterus...
-
A recipe for potato salad yields 4 gallons of potato salad. The recipe calls for 2 quarts of sour cream. How many cups of sour cream should you add if you want to make 1 gallons of potato salad?
-
Adriana wishes to accumulate $2,000,000 in 35 years. If 35 end-of-year deposits are made into an account that pays interest at a rate of 7 percent compounded annually, what size deposit is required...
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
An HNO 3 (aq) solution has a pH of 1.75. What is the molar concentration of the HNO 3 (aq) solution? a) 1.75 M b) 5.6 * 10 -13 M c) 56 M d) 0.018 M
-
Combine the reaction in eq. 3.53 with a nucleophilic substitution to devise a. A two-step synthesis of b. A four-step synthesis of CH3C¡CCH2CH3 from acetylene and appropriate alkyl halides....
-
Combine a nucleophilic substitution with a catalytic hydrogenation to synthesize a. cis-3-heptene from butyne and bromopropane. b. CH3CH2CH2OH from CH2=CHCH2Br.
-
Draw a reaction energy diagram for the reaction between CH3CH2CH2Br and sodium cyanide (NaCN). Label the energy of activation (Ea) and H for the reaction.
-
The initial margin on a GBP futures contract is $2035 and maintenance is $1850. You buy one contract (62,500 Pounds) at $1.3100 and place $2035 in your account. The price of your contract drops to...
-
1.Define a).Market research transnational firm b).Target market c).Maslow's hierarchy of needs d).ethnocentric polycentric Geocentric Explain Please create a response to the question below that is...
-
Describe one product development recommendation and one market development recommendation for Tesla. Explain why this idea should be adopted by the Tesla EV company in the New Zealand market.

Study smarter with the SolutionInn App


