Assign R or S configuration to each chirality center in the followingmolecules: (c) (a) (b) NH2
Question:
Assign R or S configuration to each chirality center in the followingmolecules:
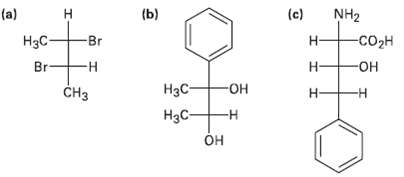
Transcribed Image Text:
(c) (a) (b) NH2 Н -соон H3C- Br н- Br -н н- он -он CHз Нэс- н- -н Нас- он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
a H3C Br H I...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to each chirality center in the following monosaccharide?s and tell whether each is a D sugar or an L sugar: (b) (a) - (c) - - - - - - - - - - CH- CH2 CH2
-
Assign R or S configuration to each chirality center in prostaglandin E2 (Figure) the most abundant and biologically potent of mammalianprostaglandin RS .CO2H Arachidonic acid PGH2 H. RS RS " " ...
-
Assign R, S configuration to each chirality center in the following molecular model of the amino acid isoleucine (blue =N):
-
In general, algorithms are classified into paradigms like: greedy, dynamic, optimization, brute-force, and divide & conquer. Each of the following phrases describes one of these paradigm 1) A...
-
Of the organizational approaches introduced in the chapter, which is best suited for writing a report that answers the following questions? Briefly explain why. a. In which market segment-energy...
-
Suppose that the silver-silver chloride electrode in Figure 14-2 is replaced by a saturated calomel electrode. Calculate the cell voltage if [Fe 2+ ] / [Fe 3+ ] = 2.5 10 -3 . Figure 14-2
-
Compare and contrast assurance and attestation services.
-
Major Funds. The Town of Trenton has recently implemented GAAP reporting and is attempting to determine which of the following special revenue funds should be classified as major funds and therefore...
-
Refer to the cost data, Picture below. Take off the square feet of wall forms and cubic yards of ready mix concrete for the walls of the elevator pit. Determine the total material and labor cost for...
-
Evaluate the function \(f(x, y)=\frac{2}{3} e^{-x / 2} \cos (y-1)\) for \(x=-0.23, y=2.7\) by: a. Using the subs command. b. Conversion into a MATLAB function.
-
Draw the three-dimensional furanose form of ascorbic acid (Problem 25.32) , and assign R or S stereochemistry to each chirality center. . c=0 Ascorbic acid - CH2
-
Draw Fischer projections of the following molecules: (a) The S enantiomer of 2-bromobutane (b) The R enantiomer of alanine, CH3CH (NH2) COOH (c) The R enantiomer of 2-hydroxypropanoic acid (d) The S...
-
Describe the basic hypothesis testing procedure.
-
American TV purchases electronic parts from Toko Corporation of Japan on account for 50,000,000 yen when the spot rate is $0.0075 on 12/12/22. On 12/31/22 the current exchange rate for yet is $.0076....
-
Based on the below data, calculate the estimated cost of the merchandise inventory on October 31 using the retail method. (round to one decimal place) Cost Retail October 1 Merchandise inventory...
-
When Armeni Toys, Inc. (ATI) receives telephone and fax orders, the billing department prepares an invoice. The invoice is mailed immediately. A copy of the invoice serves as a shipping notice and is...
-
For each risk described below identify a SPECIFIC internal control activity (not just the general category) that can be implemented in order to address the risk: a) Merchandise is sold to an...
-
Using the Ending Balances data for each account and the basic models for a Balance Sheet and Income Statement as provided below: a. Prepare the Physician's Clinic Balance Sheet for March 31, 20xx b....
-
An atom has four energy levels. How many spectral lines are seen in its emission spectrum? In its absorption spectrum? How many absorption lines?
-
Write the statement to store the contents of the txtAge control in an Integer variable named intAge.
-
What is a hydronium ion? Does H + exist in solution by itself?
-
HCFC-123 (CF3CHCl2) and HFC-134a (CF3CH2F) are replacements for chlorofluorocarbons (CFCs), which are known to deplete the stratospheric ozone layer, as described in the "A Word About...CFCs, the...
-
Predict the product from the SN2 reaction of a. Trans-4-methylcyclohexyl bromide with cyanide ion. b. (S)-2-bromopentane with cyanide ion. c. (R)-2-chlorobutane with NaSH.
-
Arrange the following compounds in order of decreasing SN2 reactivity toward sodium ethoxide: CH3 CH3 CH3CH2CHB CH CHCH2Br CH,CH2CH2CH2Br
-
What choice is a multiple of 5? a) 93 b) 63 c) 65 d) 84
-
Determine how many tablets will be needed to give the dosage. Prepare a dosage of 6.4 mg using tablets with a strength of 1.6 mg.
-
(a). Company Inc., private consumer regulator firm, claims that the standard deviation of the rating of Formula One races in the town main car racing circuit was at least 15.34. (b). An outside...

Study smarter with the SolutionInn App


