Each of the following compounds has a single 1H NMR peak. Approximately where would you expect each
Question:
Each of the following compounds has a single 1H NMR peak. Approximately where would you expect each compound toabsorb?
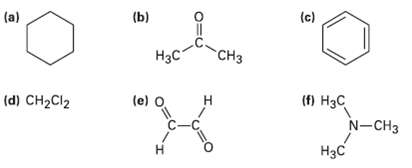
Transcribed Image Text:
(c) (b) (a) Нас "СHз (d) CH2CI2 (e) O (п Нас N-CHз н C-C Нзс н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
Compound a C6H12 b CH3COCH 3 c C6H6 d CHCl ...View the full answer

Answered By

Vineesh kumar V
To work in your esteemed organization where I can prove my expertise and work towards the growth of the organization
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Each of the following compounds has a nitrogennitrogen bond: N2, N2H4, N2F2. Match each compound with one of the following bond lengths: 110 pm, 122 pm, 145 pm. Describe the geometry about one of the...
-
Each of the following compounds has been prepared from p-nitroaniline. Outline a reasonable series of steps leading to each one. (a) p-Nitrobenzonitrile (d) 3, 5-Dibromoaniline (b) 3, 4,...
-
Each of the following compounds has been prepared from o-anisidine (o-methoxyaniline). Outline a series of steps leading to each one. (a) o-Bromoanisole (d) 3-Fluoro-4-methoxybenzonitrile (b)...
-
According to Thomson Financial, last year the majority of companies reporting profits had beaten estimates. A sample of 162 companies showed that 114 beat estimates, 29 matched estimates, and 19 fell...
-
How would you characterize the nature of competition in the restaurant industry? Are there submarkets with distinct competitive pressures? Are there important substitutes that constrain pricing?...
-
The critical temperature (K) and pressure (atm) of a series of halogenated are as follows: (a) List the intermolecular forces that occur for each compound. (b) Predict the order of increasing...
-
Differentiate between traditional and cash flow measures. Why are cash-flow-based metrics more useful for analyzing the historical and forecasted financial performance of entrepreneurial firms?
-
Cost of goods manufactured. Consider the following account balances (in thousands) for the Canseco Company: If you want to use Excel to solve this problem, go to the Excel lab at...
-
As a long-term investment, Painters' Equipment Company purchased 20% of AMC Supplies Inc's 470,000 shares for $550,000 at the beginning of the fiscal year of both companies. On the purchase date, the...
-
Australian Flying School (AFS) is a company that provides aircraft pilot training and specialist instruction services to individuals (clients) which was incorporated in the State of Victoria in 1980....
-
How many absorptions would you expect S)-malate, an intermediate in carbohydrate metabolism, to have in its NMR spectrum?Explain. (S)-Malate
-
Identify the different kinds of non-equivalent protons in the following molecule, and tell where you would expect each toabsorb. CH-CH C
-
Drake Cushing and Shawn Tadlock started the CT partnership on January 1, 2013. The business acquired $70,000 cash from Cushing and $140,000 from Tadlock. During 2013, the partnership earned $75,000...
-
Read the summary of the case Daughtrey v. Ashe on page 369 of the Cheeseman textbook (Under "Critical Legal Thinking Cases") and in an original post explain to the class whether or not you think the...
-
1. What land is particularly significant for you? It might be your current home, a previous home, a beach, bushland, a mountain etc. 2. Consider these questions in preparation for reading Alexander...
-
DISCUSSION 13: Two male juveniles and one female juvenile are out drinking together. At some point in the evening, the two young men rape the female juvenile. She has no recollection of the event....
-
In the Bombardier and Unifor Local1075 arbitration case; What obligations does the Unifor Local 1075 union have toward members of theBombardier bargaining unit? What action can a unionized employee...
-
The differences in the Cultures of Psychology and the Legal System by explaining the differences betweenrulingsanddata. One of these explanations is that precedents influence current rulings in...
-
A plane travels in a straight line from position A to position B in \(65 \mathrm{~min}\), moving at an average speed of \(400 \mathrm{~km} / \mathrm{h}\). In a car traveling from \(A\) to \(B\), the...
-
What is a manufacturing system?
-
Calculate the osmotic pressure of a solution containing 24.6 g of glycerin (C 3 H 8 O 3 ) in 250.0 mL of solution at 298 K.
-
Unlike ethylene glycol, propylene glycol (propane-1,2-diol) is nontoxic because it oxidizes to a common metabolic intermediate. Give the structures of the biological oxidation products of propylene...
-
Predict the major products of the following reactions. (a) Ethyl tosylate + potassium tert butoxide (b) Isobutyl tosylate + NaI (c) (R) 2 hexyl tosylate + NaCN (d) The tosylate of cyclohexlmethanol +...
-
Complete the following conversion table. 1700 1640 1600 400 T(cm ) A() 2.50 3.03 3.33 4.55 4000 25.0
-
Georgeland Cycles makes and sells two models of electric bicycles. The Commuter (a folding model) sells for $2,505.00 and the Tour- X (a fat-tire trail model) sells for $4,505.00. Unit variable costs...
-
How do resistance genes alter the effectiveness of antibiotics that target proteins ?
-
Able Co. received $20,000 from a customer on April 1 for services to be provided partially in April and the rest in May. At the end of April, 11% of the services were complete. What is the amount...

Study smarter with the SolutionInn App


