Identify the different kinds of non-equivalent protons in the following molecule, and tell where you would expect
Question:
Identify the different kinds of non-equivalent protons in the following molecule, and tell where you would expect each toabsorb.
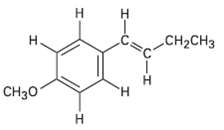
Transcribed Image Text:
н Н CH-CHз Cнзо н н ーエ т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
CHO IS H H HIC 31 5 H H 2 1 CHCH3 Proton 1 2 3 4 5 6 7 V 10 18 61 63 72 68 38 Ki...View the full answer

Answered By

Anum Naz
Lecturer and researcher with 10+ years of experience teaching courses in both undergraduate and postgraduate levels. Supervised 17 BA theses, 07 MA theses, and 1 Ph.D. dissertations. Edited and co-authored 2 monographs on contemporary trends in political thought. Published over articles in peer-reviewed journals.
4.80+
11+ Reviews
52+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify a situation where you would expect the first factor to have high loadings on all variables and to account for almost all the variance.
-
Go to www.howtoinvestigate.com. a. Identify the different kinds of background checks that investigative professionals provide. b. What information does a basic background investigation provide? c....
-
Discuss the different kinds of incentives that can be used to motivate employees to become involved in quality improvement programs. Explain gain sharing.
-
Companies use off-balance sheet accounting so that they do not have to include certain assets and liabilities in their financial statements. Off-balance sheet accounting is often used to make the...
-
In a recent antitrust case, it was necessary to determine whether certain elite schools (mainly the Ivy League schools and MIT) constituted a separate market. How would you go about identifying the...
-
Using this graph of CS2 data, determine (a) the approximate vapor pressure of CS2 at 30 °C, (b) the temperature at which the vapor pressure equals 300 torr, (c) the normal boiling point of CS2?...
-
Discuss the advantages and disadvantages of the various crowdfunding models.
-
A. J. Johnson & Co. recorded certain revenues on its books in 2011 and 2012 of $15,400 and $16,600, respectively. However, such revenues were not subject to income taxation until 2013. The company...
-
Bill Jorgenson lives in New York and is terminally ill. Suppose Bill's son, Harold, convinces his father that Cheryl Jorgenson, Bill's wife and Harold's mother, is initiating institutional commitment...
-
Determine Zi Zo and Av for the network of Fig. 8.84. +20 V aSS 12 mA 3 91 = 45 k Z 10 MS2
-
Each of the following compounds has a single 1H NMR peak. Approximately where would you expect each compound toabsorb? (c) (b) (a) "H (d) CH2CI2 (e) O ( N-CH C-C
-
How many peaks would you expect in the 1H NMR spectrum of 1, 4-dimethyl-benzene para-xy1ene or p-xylene)? What ratio of peak areas would you expect on integration of the spectrum? Refer to Table 13.3...
-
What is the difference between RER and SER? SER is not very common in plants, but several examples are given in the text. What are they?
-
Suppose EG 3, EB=8, AF=6, mZEBG=23, mZEGF 28, and m/CAE m/CAF= 52. Find m/CAF. A nswer. Do not include the degree symbol in your answer) F G B
-
Based on Victoria THOMPSON individually and on behalf of Decedent Russell Gene Thompson, Plaintiff,v.UNITED STATES of America, Defendant. 1. Summarize the facts, identify the parties, provide the...
-
Case: Tomasella v. Nestl USA, Inc CREATE A POSTER FOR THE PRESENTATION CREATE A POSTER FOR THE PRESENTATION CREATE A POSTER FOR THE PRESENTATION INCLUDE PICTURES Introduction 3 main points of the...
-
Watch this news clip: https://www.pbs.org/newshour/show/justices-weigh-religion-discrimination-and-dignity-in-colorado-wedding-cake-case And answer the following questions. Whose position do you...
-
In addition to understanding budgeting processes, is it important to know the entity that prepared a budget for a criminal justice, Texas department of Criminal Justice correctional services? Why or...
-
A child rides her bike five blocks east and then three blocks north. It takes her \(15 \mathrm{~min}\), and each block is \(160 \mathrm{~m}\) long. What are \((a)\) the magnitude of her displacement,...
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
An aqueous solution containing 35.9 g of an unknown molecular (nonelectrolyte) compound in 150.0 g of water has a freezing point of -1.3 C. Calculate the molar mass of the unknown compound.
-
Ethers are not easily differentiated by their infrared spectra, but they tend to form predictable fragments in the mass spectrum. The following compounds give similar but distinctive mass spectra....
-
Account for the peaks at m/z 87, 111, and 126 in the mass spectrum of 2,6-dimethylheptan-4-ol. 100 OH 80 2,6-dimethylheptan-4-o1 E 40 83 111 126 20 10 20- 30 40 50 60mm 80 m90ml00 110T20 130T 140...
-
Predict the characteristic infrared absorptions of the functional groups in the following molecules. (a) Cyclohexene (b) Pentan-2-ol (c) Pentan-2-one (d) Pent-1-yne (e) Diethylamine (f) Pentanoic...
-
The following equation of state is valid for a given pure gas: Vi = RT P - RT T [0,3 - 6.5 10-4] Where T is in K, P is in kPa, and the term within square parentheses () has units of 1/kPa. Develop an...
-
How do sociological theories of deviance and social control elucidate the processes through which individuals and groups are labeled as "deviant" and subjected to stigmatization or criminalization ?
-
Current Attempt in Progress The following information relates to Cullumber Company's accounts receivable Accounts receivable balance on December 31, 2023 $948,000 Allowance for expected credit losses...

Study smarter with the SolutionInn App


