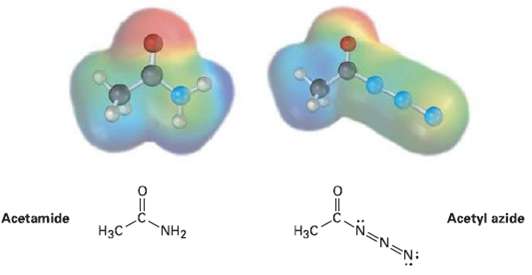
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which
Question:
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more reactive in nucleophilic acyl substitution reactions? Explain.
Transcribed Image Text:
Acetamide Acetyl azide NH2 Нзс Нас
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
According to the electrostatic potential maps the carbonyl carbon of ac...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which store do you think is more expensive-physical or online? A recent survey (USA Today, December 10, 2012, p. 1B) found that 46% of people aged 20 to 40 thought that physical stores were more...
-
Which of the alkyl halides is more reactive in an E2 reaction? a. CH3CH2CH2Br or b. c. d. CH,CH2CHCH3 Cl or CH cHy CH3CHCH2CHCH or CH3CH2CH2CCH3 Br Br CH, CH CH3 CH3
-
Which company and business model do you think is most likely to dominate the Internet and why ?
-
Give the approximate temperature at which creep deformation becomes an important consideration for each of the following metals: nickel, copper, iron, tungsten, lead, and aluminum.
-
What are detractors saying about PowerPoint, and why are they condemning it? Can you present a counterargument?
-
In week 1, the "low price" manager cuts prices by 50%. In week 2, the "high price" manager raises prices by 50%, and so forth. Suppose an item started out at $100. A store has two managers, one who...
-
Outline the project planning process based on identified resources.
-
Eleby Company has gathered the following information. Units in beginning work in process .. 0 Units started into production ... 36,000 Units in ending work in process .... 6,000 Percent complete for...
-
10. Rewrite the given logarithmic expressions: a. 27x3 1083 Vy b. log, x-3logy ---log, z 2
-
Chick-fil-A Create a 10- to 12-slide presentation with detailed speaker notes and visuals on every slide that includes the following elements: An analysis of the organizations current culture (e.g.,...
-
The following structure represents a tetrahedral alkoxide-ion intermediate formed by addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the...
-
Give IUPAC names for the following compounds: o (c) CHH2H2 CH2CH3 (a) (b) CH3CH2CHCHI NH2 (d) (e) (f) CH3CHCH,NHCH3 CH-CH-C H CH Br (h) (g) SCH(CH3)2
-
The board of trustees of a local church has asked for your help with the controls activities for the offering collections made at weekly services. At a board meeting, you learn the following: 1. The...
-
For the equation 2x+14=-2, x equals
-
Express -36 in terms of i: -36 =
-
0.042 0.0096 Simplify the expression using scientific notation 0.84 x 0.012
-
1. Use algebraic methods and properties of limits to evaluate x2 - 4 lim x-2x+2
-
Divide. (2x+6x-13)+(x+2)
-
In what way is the silicate tetrahedron like a building block or a Lego?
-
Accounting policies and practices that are most important to the portrayal of the companys financial condition and results, and require managements most difficult, subjective, or complex judgments...
-
Write orbital diagrams (boxes with arrows in them) to represent the electron configuration of carbon before and after sp 3 hybridization.
-
Give the structure that corresponds to each of the following molecular formulas and NMR spectra: (a) C5H10; 1.5, s (b) C2H2F3I: 3.56 (q, J = 10 Hz) (c) C6H14O: 0.91 (6H, d, 7 = 7 Hz); 1.17(6H,...
-
A compound A reacts with H2 over Pd/C to give methylcyclohexane. A colleague, A1 Keen, has deduced that the compound must be either 1-methylcyclohexene or 3-methylcyelohexene. You have been called in...
-
A compound A reacts with H2 over Pd/C to give methylcyclohexane. A colleague, A1 Keen, has deduced that the compound must be either 1-methylcyclohexene or 3-methylcyelohexene. You have been called in...
-
Privatizing Social Security in the United States has been a topic of debate for many years, with strong opinions on both sides since its creation as part of President Franklin D. Roosevelt's New...
-
Economists measure the price level by using a basket of goods and services and calculating how the total cost of buying that basket of goods will increase over time. Economists often express the...
-
Diverse Perspectives on the World Economy assignment You are required to consider a contemporary economic or social issue that is currently attracting media attention. It could be a local, national,...

Study smarter with the SolutionInn App


