Give IUPAC names for the following compounds: o (c) CHH2H2 CH2CH3 (a) (b) CH3CH2CHCHI NH2
Question:
Give IUPAC names for the following compounds:
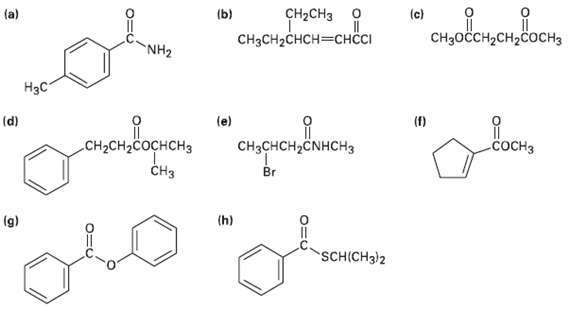
Transcribed Image Text:
oн (c) CHзоссH2сH2соснз CH2CH3 о (a) (b) CH3CH2CHCH—снссI NH2 Нзс (d) (e) (f) CH3CHCH,ČNHCH3 CH-CH-Cоснснз сосHз CHз Br (h) (g) SCH(CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (22 reviews)
a d H3C pMethylbenzamide g NH Isopropyl 3phenylpropanoate b CH3...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following compounds: CH CH3CH2CH=CCH2CH3 (b) (a) CH CHCH-H CHCH2CH2CHCHCHCH2CH3 CH CH (d) (c) CHCHCHCHC CH
-
Give IUPAC names for the followingcompounds: H CHCH2CCH H CH CHH2CH2CH (b) (e) CHH2H2C CH (a) H-H H CH3CH2CHCH2CH,CHCH3 (e) CH H CHCH2CH2CHCH2CH CH CH-H>H3 (d)
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Compare sand, die, investment, lost foam, and continuous casting techniques.
-
"Communicate-don't decorate." This principle is one of 20 rules that graphic designer and educator Timothy Samara discusses in his book Design Elements: A Graphic Style Manual. How could you apply...
-
Using the histograms (from Section 6.6, Exercises 9-12), estimate the median, the mode, and the expectation. 1. 2. 3. 4. 0.25 r 0.2 0.15 0.1 0.05 0 2 46 10 Measurement 0.3 0.25 0.2 0.15 E 0. 0.05 0...
-
Evaluate project feasibility in terms of resources, funding, and facilities.
-
The following facts pertain to a noncancelable lease agreement between Lennox Leasing Company and Gill Company, a lessee. Inception date: .................. May 1, 2012 Annual lease payment due at...
-
Solve each of the following: 14. 23x-1= 8 15. 16. 3x-4 27 log3 (2x+3)=2
-
Given (a) Find the vector projection p of x onto y. (b) Verify that x - p is orthogonal to p. (c) Verify that the Pythagorean Law holds for x, p, and x - p, that is, ||x|| = ||P||2 + ||x - p||2 2120...
-
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more reactive in nucleophilic acyl substitution reactions?...
-
Draw structures corresponding to the following names: (a) p-Bromophenylacetamide (b) m-Benzoylbenzamide (c) 2, 2-Dimcthylhcxanamidc (d) Cyclohexyl cyclohexanecarboxylale (e) Ethyl...
-
Who developed the audit data standards? In your opinion, why is it the right group to develop and maintain them rather than, say, the Big 4 firms or a small practitioner?
-
Graph the polynomial function g(x)=-x(x-1)(x+4)
-
The digits 1, 2, 3, 4, and 5 are randomly arranged to form a five-digit number. Complete parts (a) and (b) below. (a) Find the probability that the number is odd. The probability that the number is...
-
Y 3. Expand Y x ( % 1 ) ( 2 + 3 )
-
Express -121: -121 in terms of i: =1
-
Solve for y. 2-2+15=39
-
How is coal unlike other sedimentary rocks?
-
A company has the following incomplete production budget data for the first quarter: In the previous December, ending inventory was 200 units, which was the minimum required, at 10% of projected...
-
Write orbital diagrams (boxes with arrows in them) to represent the electron configurationswithout hybridizationfor all the atoms in SF 2 . Circle the electrons involved in bonding. Draw a...
-
How many absorptions should be observed in the 13C NMR spectrum of each of the following compounds? (Assume that the chair interconversion is rapid.) CH3 H,C
-
Explain how the proton NMR spectra of the compounds within each of the following sets would differ, if at all. (CH)2CHC and (CH CDCI
-
Although this chapter has discussed only nuclei that have spin 1/2, several common nuclei such as 14N and deuterium (2H, or D) have a spin of 1. This means that the spin has three equally probable...
-
Daniel made the following transactions during the first week of January. 1 January Buys stock for 350 on credit 3 January Makes sales of 740 on credit 4 January Cash paid to a supplier of 120, for...
-
Please answer all parts of the following question. Given the following information: Total Product (TP) Price Fixed Cost (TFC) Variable Cost (TVC) 0 $1000 $600 $ 0 1 1000 600 1200 2 1000 600 2000 3...
-
Imagine you are an analyst working at a financial consulting firm. Your organization has been hired by UBS to provide advice following a major legal and ethical crisis. You have been asked to draft...

Study smarter with the SolutionInn App


