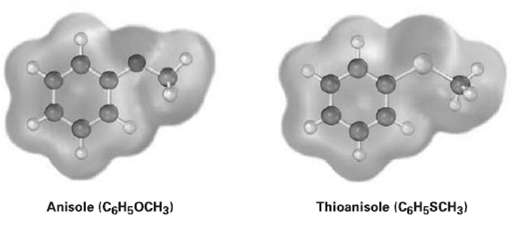
Electrostatic potential maps of anisole and thioanisole are shown. Which do you think is the stronger acid,
Question:
Electrostatic potential maps of anisole and thioanisole are shown. Which do you think is the stronger acid, p-methoxybenzoic acid or p-(methylthio) benzoic acid?Explain.
Transcribed Image Text:
Anisole (CGH5OCH3) Thioanisole (C6H5SCH3)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The electrostatic potential maps show that the aromatic ring of anisole ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which do you think is more important for organizations: downward communication or upward communication?
-
Which do you think is more important for organizations: formal or informal communication?
-
Which do you think are more important for an international marketer who is introducing a new product, a push or a pull strategy? Why? What specific types of tactics do you think are most important in...
-
You name the catastrophe, and JIT has been through it and survived. Toyota Motor Corporation has had its world-renowned JIT system tested by fire. The massive fire incinerated the main source of...
-
Jonas and Rande are facing a difficult situation. What sources of conflict contributed to the problems they are having?
-
The graph below shows supply and demand curves for a new mp3 player accessory. a. What is the equilibrium price? b. Describe the relationship of supply and demand if the item were sold for $20. c....
-
What System Elements comprise a systems Physical Environment domain?
-
A few years back, Dave and Jana bought a new home. They borrowed $230,415 at a fixed rate of 5.49% (15-year term) with monthly payments of $1,881.46. They just made their twenty-fifth payment and the...
-
Rem Starr, managing director of Irmo Manufacturing, is evaluating the viability of installing a new machine to expand operations. The machine under consideration costs $200,000. It is anticipated the...
-
The Griffins were driving in a blizzard when a truck hit them as it was sliding off the road. The truck driver received minor injuries, but Wanda and Frank died at the scene of the accident. Amy,...
-
The following carboxylic acid can?t be prepared from an alkyl halide by either the nitrile hydrolysis route or the Grignard carboxylation route. Explain.
-
Give IUPAC name for the followingcompounds: (c) NC. CH 2 () (a) CHCH2CH2CH CH2 CH () CH CH2CO2H (d) (e) CHH2H2H2CH3 CHCN CH (g) Br (h) CN BRCH2CHCH2CH2CO2H
-
A curve has equation a. Find dy/dx and d 2 y/dx 2 in terms of x. b. Find the coordinates of the stationary point on the curve and determine its nature. c. Find the volume of the solid formed when the...
-
How do power dynamics within institutions influence the manifestation of integrity among their members, and what strategies can be employed to mitigate integrity breaches in hierarchical structures ?...
-
Weighted Average Shares At the beginning of 2 0 1 9 , Hardin Company had 3 4 0 , 0 0 0 shares of $ 1 0 par common stock outstanding. During the year, it engaged in the following transactions related...
-
Discuss how two magazine advertisements for the same product (such as a health and beauty product) would be different if one is based on the CPM perspective of consumer information processing and the...
-
Think of a company that has built its business solely on a digital realm. How has it successfully or unsuccessfully built a foundation of trust with its consumers? How has technology enabled the...
-
Consider the following: Net Income Depreciation Expense Gain on Sale of Land Increase in Inventory Increase in Wages Payable Payment of Dividends $51,900 36,000 22,500 6,150 18,450 6,000 Calculate...
-
We happen to find that the ground state of the harmonic oscillator (and the coherent states in general) is a minimum uncertainty state, but can we do the converse? That is, can we directly determine...
-
Determine whether the lines are parallel, perpendicular, or neither. 2x + 3y = -12, 2y - 3x = 8
-
One kind of polyester is a condensation copolymer formed from terephthalic acid and ethylene glycol. Draw the structure of the dimer. HO Terephthalic acid OH HD-CH-CH-OH Ethylene glycol
-
Direct oxidation of an aldose affects the aldehyde group first, converting it to a carboxylic acid, and most oxidizing agents that will attack 1o alcohol groups will also attack 2o alcohol groups....
-
Write three-dimensional formulas for each aldotetrose and ketopentose isomer in Practice Problem 22.1 and designate each as a d or l sugar.
-
Give appropriate structural formulas to illustrate each of the following: (a) An aldopentose (b) A ketohexose (c) An l-monosaccharide (d) A glycoside (e) An aldonic acid (f) An aldaric acid (g) An...
-
On January 1, 2023, Bertrand, Incorporated, paid $86,900 for a 40 percent interest in Chestnut Corporation's common stock. This investee had assets with a book value of $229,500 and liabilities of...
-
Landen Corporation uses a job-order costing system. At the beginning of the year, the company made the following estimates: Direct labor-hours required to support estimated production Machine-hours...
-
Explain the concept of gene-environment interactions, and how these interactions influence the expression of complex traits and the risk of developing multifactorial diseases ?

Study smarter with the SolutionInn App


