One kind of polyester is a condensation copolymer formed from terephthalic acid and ethylene glycol. Draw the
Question:
One kind of polyester is a condensation copolymer formed from terephthalic acid and ethylene glycol. Draw the structure of the dimer.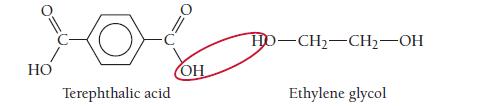
Transcribed Image Text:
HO Terephthalic acid OH HD-CH₂-CH₂-OH Ethylene glycol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The polyester named Lactomer® is an alternating copolymer of lactic acid and glycolic acid. Lactomer is used for absorbable suture material because stitches of Lactomer hydrolyze slowly over a...
-
Glyptal resin makes a strong, solid polymer matrix for electronic parts. Glyptal is made from terephthalic acid and glycerol. Draw the structure of Glyptal, and explain its remarkable strength and...
-
Ethylene glycol is used as an automobile antifreeze and in the manufacture of polyester fibers. The name glycol stems from the sweet taste of this poisonous compound. Combustion of 6.38 mg of...
-
Both high-income and low-income employees are covered by cafeteria plans. Under such plans, all employees may select from a list of non-taxable fringe benefits or they may elect to receive cash in...
-
On December 23, Wyman, a lawyer representing First National Bank & Trust (defendant), wrote to Zeller (plaintiff) stating that he had been instructed to offer a building to Zeller for sale at a price...
-
Paul Larson says that people tend to join a company but leave their supervisor. Do you agree or disagree? Explain your answer.
-
Design Data sold a piece of machinery to HHB Drafting Company. However, after HHB had taken possession of the machine, it discovered damage and revoked the contract. The court found that the...
-
Hearne Company has a number of potential capital investments. Because these projects vary in nature, initial investment, and time horizon, management is finding it difficult to compare them. Project...
-
4. (7 Marks) ROVNO Ltd. just invested $65,000 into a new local network. Services provided by the network will bring $27,000 per year in the next five years. The following information is given: -...
-
Nomex, a condensation copolymer used by firefighters because of its flame-resistant properties, forms from isophthalic acid and m-aminoaniline. Draw the structure of the dimer. HO Isophthalic acid OH...
-
Saran, the polymer used to make Saran Wrap, is an addition polymer formed from two monomersvinylidene chloride and vinyl chloride. Draw the structure of the polymer. H C=C Cl H Cl Vinylidene chloride...
-
Write code that does the following: opens a file named MyName.txt, writes your first name to the file, and then closes the file.
-
Explain about the application software available for Unix operating system?
-
1. Provide a brief description of your implicit theory of leadership, i.e., your beliefs about the characteristics and behaviors of effective leaders. Does this description seem to agree with or be...
-
John Kotter describes "What leaders really do." Peter Drucker describes "What makes an Effective Executive." What are the similarities, if any, between the two theories? What are the differences, if...
-
Explain various mapping procedures of cache memory with an example.?
-
What is cache memory? How is it different from virtual memory?
-
On January 1 Revis Consulting enters into a contract to complete a cost reduction program for Green Financial over a six-month period. Green will pay Revis $20,000 at the end of each month. If total...
-
Calculate I, , and a for a 0.0175 m solution of Na 3 PO 4 at 298 K. Assume complete dissociation. How confident are you that your calculated results will agree with experimental results?
-
(Challenging) Building on the concept of equipartition, demonstrate that for any energy term of the form ax 2 , where α is a constant, the contribution to the internal energy is equal...
-
For a system of energy levels, m = m 2 , where is a constant with units of energy and m = 0, 1, 2, , . What is the internal energy and heat capacity of this system in the high-temperature limit?
-
What is the contribution to the internal energy from translations for an ideal monatomic gas confined to move on a surface? What is the expected contribution from the equipartition theorem?
-
You do not need to understand how the graphical user interface is developed nor need you to make any changes in the graphical interface Make following changes. L 2. Convert the interface and classes...
-
Using C Programming for the following: Run the program below with the following values: #include int main(void) { int x, y, p, q; printf("Enter x and y:"); scanf("%4d%5d", &x, &y);...
-
Testing & Submission Use the Compile and Run buttons below to compile and run your program in the terminal. COMPILE RUN Use the terminal to test your program, making sure all program specifications...

Study smarter with the SolutionInn App


