Epoxides are reduced by treatment with lithium aluminum hydride to yield alcohols. Propose a mechanism for thisreaction.
Question:
Epoxides are reduced by treatment with lithium aluminum hydride to yield alcohols. Propose a mechanism for thisreaction.
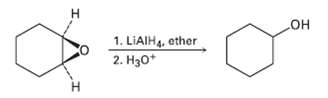
Transcribed Image Text:
н 1. LIAIH4, ether 2. Hзо* н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
0 H3O 110 Protonation H Attack of hydrid...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Epoxide?s react with Grignard reagents to yield alcohols propose a mechanism. 1. CHMgBr 2. * CH
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
Propose a mechanism for each reaction. (a) (b) Ht H3C CH3 OH H SOA Ph Ph Ph Ph
-
When considering banning a toxic chemical, why should replacement substances or processes be part of the decision?
-
What are the big differences in the way communities approach development today compared to 1990, when Porter wrote The Competitive Advantage of Nations?
-
Using the table of percentage points of the x2 distributions, find: (a) x2.05s with d.f. = 6. (b) x2.025 with df- = 22. (c) The lower .05 point with d.f. = 6. (d) The lower .025 point with d.f. = 22.
-
Why does a worker allocate his or her time over the life cycle so as to work more hours in those periods when the wage is highest? Why does the worker not experience an income effect during those...
-
On June 1, 20Y6, Hannah Ellis established an interior decorating business, Whitworth Designs. During the month, Hannah completed the following transactions related to the business: June 1. Hannah...
-
How many standards are included in the ELP accreditation standards? What stands out to you about the different areas? What are the topic areas under administration and leadership? What are the...
-
A production process consists of a three- step operation. The scrap rate is 10 percent for the first step and 6 percent for the other two steps. a. If the desired daily output is 450 units, how many...
-
Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol). CH2CH=CH2 Safrole
-
Show the structure and stereochemistry of the alcohol that would result if 1, 2-epoxycyclohexane (Problem 18.39) were reduced with lithium aluminum deuteride, LiAID4.
-
What are the first steps in performing a subdivision survey?
-
If you had limited funds to build a large telescope, which type would you choose, a refractor or a reflector? Why?
-
Greeter Soft is a private firm that was started by Sam Sung for developing software for greeting card companies. Sam had invested $200,000 in the company. One year later a venture firm Big Bucks...
-
Erin visited a farm with her mother. As they entered the barn, a cat passed by and Erin said "me pet cat." Later, as they were leaving, she said "bye-bye cat," even though the cat was no longer...
-
3. Construct a test set that is adequate with respect to LCSAJ coverage criterion for the following program segment. (40 points) 1 2 public static boolean has At Least 3 Letters (String new Password)...
-
How do lenders calculate borrowers' ability to repay their mortgage loans?
-
Meura Bancorp, a US bank, has an equity capital ratio for financial assets of 12%. Meuras strategic plans include the incorporation of additional debt in order to leverage earnings since the current...
-
Refer to the data in QS 10-1. Based on financial considerations alone, should Helix accept this order at the special price? Explain.
-
The Ocean Thermal Energy Conversion (OTEC) project in Hawaii produces electricity from the temperature difference between water near the surface of the ocean (about 27 C) and the 600 m deep water at...
-
Write resonance structures for the azide ion, N3-. Explain how these resonance structures account for the fact that both bonds of the azide ion have the same length.
-
Write structural formulas of the type indicated: (a) Bond-line formulas for seven constitutional isomers with the formula C4H10O; (b) Condensed structural formulas for two constitutional isomers with...
-
Write the Lewis structure of (a) CH2Fl2 (difluoromethane) (b) CHCl3 (chloroform).
-
Choose from the following topics to create discussion: Humanity and the Natural Environment Housing and Home Furnishings Privacy and Socializing
-
On December 31, 2020, Pearl Company had $1,101,000 of short-term debt in the for January 21, 2021, the company issued 22,800 shares of its common stock for $42 per brokerage fees and other costs of...
-
During your interview process for the position of program manager for Booker, Tee, and How you are asked to explain the relationship between cost, schedule, and performance? what is your answer to...

Study smarter with the SolutionInn App


