Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis
Question:
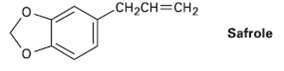
Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol).
Transcribed Image Text:
CH2CH=CH2 Safrole
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
OH OH 2 NaH O ...View the full answer

Answered By

Shyamsunder K
Teaching my kids and students ( though I am not a teacher by profession) around me and helping them in easily comprehend the subjects; though teaching is my passion but not as profession and students who I have taught have achieved success in taking the exams confidently and perform great;y.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Karahanaenone, a terpenoid isolated from oil of hops, has been synthesized by the thermal reaction shown. Identify the kind of pericyclic reaction, and explain how karahanaenone isformed. CH CH eat...
-
Safrole is contained in oil of sassafras and was once used to flavor root beer. A 2.39-mg sample of safrole was dissolved in 103.0 mg of diphenyl ether. The solution had a melting point of 25.70C....
-
Propose a formula for hydrogen peroxide, a substance used as a bleaching agent. (Curiously, this compound does not behave as an acid, despite its formula. It behaves more like a classic...
-
The following financial statements for Brownstone plc are a slightly simplified set of published accounts. Brownstone plc is an engineering business that developed a new range of products in 2007....
-
Evaluate how Apple can gain business intelligence through the implementation of a customer relationship management system.
-
Describe several structural decisions that a low-cost hotel such as a Days Inn would have to make. Describe infrastructural decisions that such a hotel would make. How would the decisions made in...
-
In connection with his examination of the financial statements of the Olympia Manufacturing Company, a CPA is reviewing procedures for accumulating direct labor hours. He learns that all production...
-
Andrew-Carter, Inc. (A-C), is a major Canadian producer and distributor of outdoor lighting fixtures. Its products are distributed throughout South and North America and have been in high demand for...
-
In a 2D animation, a ball initially located at P = (5,5) and travelling with velocity v = (-2,-3) hits the surface defined by points Q = (2,4) and R = (6,1). The ball then reflects from the surface....
-
Drinkwater Inc. reported the following information: Other information: 1. No shares were issued during the years ended March 31, 2020 and 2019. 2. No dividends were declared or paid during the years...
-
Meerwein?s reagent, triethyloxonium tetra-fluoroborate, is a powerful ethylating agent that converts alcohols into ethyl ethers at neutral pH. Show the reaction of Meerwein?s reagent with...
-
Epoxides are reduced by treatment with lithium aluminum hydride to yield alcohols. Propose a mechanism for thisreaction. 1. LIAIH4, ether 2. H*
-
Which are true of a JAR file that has only one module-info.class file, placed in the META-INF directory? (Choose two.) A. It is an automatic module if on the classpath. B. It is an automatic module...
-
Ted is 56 and has determined that he will need retirement income equal to $50,000/year in today's dollars. He plans to retire in 10 years. He assumes that inflation will average 3% per year.Ignoring...
-
The significance of an event to a particular financial market or stock can be determined through changes in the exchange index or changes in individual stock prices. Explain how to ensure that the...
-
At December 31, 2020, the assets of Mandarin Company's cash-generating unit (CGU) that include goodwill are shown below (in millions) on the company's statement of financial position: ($ in millions)...
-
Carrington Co. operates its two divisions as investment centers. Information about these divisions follows. Division 1 Division 2 Sales $3,120,000 $1,110,000 Total variable costs 1,578,000 198,000...
-
Prepare an income statement for the year through gross profit. The following information is available for Sheridan Company at December 31, 2022: 1. Inventory balance Beginning of Year End of Year...
-
Emily Stansbury is a licensed dentist. During the first month of the operation of her business, the following events and transactions occurred. Emily uses the following chart of accounts: No. 101...
-
On the basis of the details of the following fixed asset account, indicate the items to be reported on the statement of cashflows: ACCOUNT Land ACCOUNT NO. Balance Date Item Debit Credit Debit Credit...
-
A tank contains 20 percent liquid water and 80 percent steam by volume at 200 C. Steam is withdrawn from the top of the tank until the fluid remaining in the tank is at a temperature of 150 C....
-
Consider another chemical species like the ones in the previous problems in which a carbon atom forms three single bonds to three hydrogen atoms but in which the carbon atom possesses a single...
-
Draw a three-dimensional orbital representation for each of the following molecules, indicate whether each bond in it is a s or p bond, and provide the hybridization for each non-hydrogen atom. (a)...
-
Ozone (O3) is found in the upper atmosphere where it absorbs highly energetic ultraviolet (UV) radiation and thereby provides the surface of Earth with a protective screen. One possible resonance...
-
Implement the definition of the function rotate ToLeft(), member of the DArray class, so that it rotates all the elements of the array object to the left by one position. Example: [6, 2, 5, 3] [2, 5,...
-
Write the definition of the of the function is Full() as a member function of the DArray class. The function returns true if the array is full, or false otherwise. Functions.cpp 1 #include "DArray.h"...
-
Write a program that will calculate the trajectory of a golf ball thrown with a velocity of 25.6 m/s at an angle of 0 = 35 relative to the horizontal, from a hill that is 2.1 meters above the ground...

Study smarter with the SolutionInn App


