Epoxy adhesives arc cross-linked resins prepared in two steps. The first step involves S N 2 reaction
Question:
Epoxy adhesives arc cross-linked resins prepared in two steps. The first step involves SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin to form a low-molecular-weight prepolymer. This prepolymer is then ?cured? into a cross-linked resin by treatment with a triamine such as H2NCH2CH2NHCH2CH2NH2
(a) What is the structure of the prepolymer?
(b) How does addition of the triamine to the prepolymer result in cross-linking?
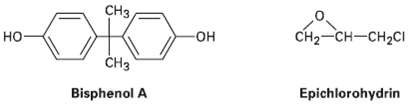
Transcribed Image Text:
CH3 CH2-CH-CH2CI но- HO- ČH3 Bisphenol A Epichiorohydrin
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
a CICHCHCH Sio o CHCHCHO 8 HCCHCHO CH3 I CH3 CH3 CH3 ...View the full answer

Answered By

Atuga Nichasius
I am a Highly skilled Online Tutor has a Bachelor’s Degree in Engineering as well as seven years of experience tutoring students in high school, bachelors and post graduate levels. I have a solid understanding of all learning styles as well as using asynchronous online platforms for tutoring needs. I individualise tutoring for students according to content tutoring needs assessments.
My strengths include good understanding of all teaching methods and learning styles and I am able to convey material to students in an easy to understand manner. I can also assists students with homework questions and test preparation strategies and I am able to help students in math, gre, business , and statistics
I consider myself to have excellent interpersonal and assessment skills with strong teaching presentation verbal and written communication
I love tutoring. I love doing it. I find it intrinsically satisfying to see the light come on in a student's eyes.
My first math lesson that I taught was when I was 5. My neighbor, still in diapers, kept skipping 4 when counting from 1 to 10. I worked with him until he could get all 10 numbers in a row, and match them up with his fingers.
My students drastically improve under my tutelage, generally seeing a two grade level improvement (F to C, C to A, for example), and all of them get a much clearer understanding!
I am committed to helping my students get the top grades no matter the cost. I will take extra hours with you, repeat myself a thousand times if I have to and guide you to the best of my ability until you understand the concept that I'm teaching you.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin forms a ?prepolymer,? which is then ?cured? by treatment with a triamine such as H 2...
-
A rod of weight W and uniform cross section is bent into a quarter circle and is supported as shown. Determine the bending moment at point J when θ = 30°.
-
A first step in regression analysis often involves developing a scatter plot of the data. Develop the scatter plots of all the possible pairs of variables, and with a brief statement indicate what...
-
Bruceton Farms Equipment Company had goodwill valued at $80 million on its balance sheet at year-end. A review of the goodwill by the company's CFO indicated that the goodwill was impaired and was...
-
How do the five forces of competition in an industry affect its profit potential? Explain. Discuss.
-
DNA renaturation curves occasionally show three distinct phases of renaturation. In this graph, DNA renaturation is plotted against "C0t" (initial concentration times time of renaturation-essentially...
-
When the heat capacity ratio is 1.67 , then the ideal gas is (a) Diatomic (b) Monatomic (c) Triatomic (d) None of these.
-
As inventory manager, you must decide on the order quantity for an item that has an annual demand of 2,000 units. Placing an order costs you $20 each time. Your annual holding cost, expressed as a...
-
What are the differences between a finance lease and an operating lease? Why is the cost of replacement tires being ignored in the lease v buy analysis? Tori's textbook states that when making a...
-
The performance (in thousands of dollars) of Sandpiper Airlines for the most recent year is shown in the following table: The static budget had been based on a budget of $.35 revenue per passenger...
-
Melmac, a thermosetting resin often used to make plastic dishes, is prepared by heating melamine with formaldehyde. Look at the structure of Bakelite shown in Section 31.5, and then propose a...
-
The polyurethane foam used for home insulation uses methane diphenyl-diisocyanate (MDI) as monomer. The MDI is prepared by acid-catalyzed reaction of aniline with formaldehyde, followed by treatment...
-
In your opinion, what is the meaning of responsible conduct with respect to the use of social media for screening purposes?
-
Gaga reported the following: Units: 656 Sales $6737 Variable Costs $620 Fixed Costs $538 If the company reduces its selling price by $1.07 per unit to generate more sales AND expects the number of...
-
Titanium Blades refines titanium for use in all brands of razor blades. It prepared a static budget for the sales of 5,000 units. These variances were observed: Actual Results and Variances,...
-
How does urbanization contribute to the acceleration of climate change, and what adaptive measures can cities implement to enhance resilience against climate-related risks ?
-
Teecorp Company provides the following ABC costing information: Activities Total Costs Activity-cost drivers Labor $320,000 8,000 hours Gas $36,000 6,000 gallons Invoices $40,000 2,500 invoices Total...
-
How do patterns of urbanization affect global migration trends, and what are the socio-political ramifications of this interrelationship ?
-
A beetle that has an inertia of \(4.0 \times 10^{-6} \mathrm{~kg}\) sits on the floor. It jumps by using its muscles to push against the floor and raise its center of mass. (a) If its center of mass...
-
A woman at a point A on the shore of a circular lake with radius 2 mi wants to arrive at the point C diametrically opposite on the other side of the lake in the shortest possible A time. She can walk...
-
When pure hydrogen iodide gas enters an evacuated cylinder, the following reactions may occur: (Note that since Gibbs energies of formation data are available for iodine in both the gaseous and solid...
-
Even though the methyl group occupies an equatorial site, the conformation shown is not the most stable one for methylcyclohexane. Explain.
-
Which of the structures shown for the axial conformation of methylcyclohexane do you think is more stable, A or B? Why? A
-
Which do you expect to be the more stable conformation of cis-1,3-dimethylcyclobutane, A or B? Why? CH3 CH3 CH3
-
Simplify. 9u6x4 4 3 3wx +9x
-
Majesty Company uses target costing to ensure that its products are profitable. Assume Majesty is planning to introduce a new product with the following estimates: Annual demand Estimated market...
-
The side of a festival food tent is staked with two guidelines as shown. If the force in the rope going from A to B is 450 lbs and the force in the rope going from A to C is 300 lbs, what is the...

Study smarter with the SolutionInn App


