Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with
Question:
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin forms a ?prepolymer,? which is then ?cured? by treatment with a triamine such as H2NCH2CH2NHCH,CH2NH2,. Draw structures to show how addition of the triamine results in strengthening he polymer. Amines are good nucleophiles and can open epoxide rings in the same way other bases can.
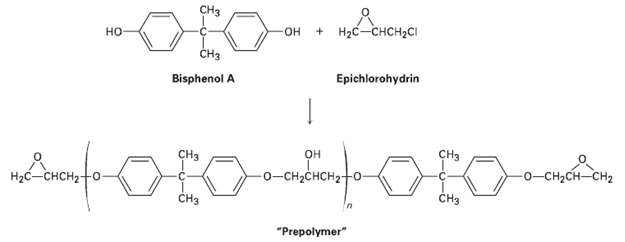
Transcribed Image Text:
CH3 Аоне OLO- + Hас-снсн-с но -он ČH3 Epichlorohydrin Bisphenol A CHз CHз н Hас — снсн?- -о-сн-снсH2 -о-сн-сH—сH2 CHз CHз "Prepolymer"
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Addition of the triamine causes ...View the full answer

Answered By

Talha Talib
I am a member of IEEE society. As i am a student of electrical engineering badge 17 but beside of this i am also a tutor in unique academy. I teach calculus, communication skills, mechanics and economics. I am also a home tutor. My student Muhammad Salman Alvi is a brilliant A-level student and he performs very well in academics when i start to teach him. His weak point was mathematics but now he is performing well in mathematics. I am a scholarship holder in Fsc as i scored 1017 marks in metric out of 1100. Later on i got scholarship in Punjab Group of Colleges. I got 2nd position in robotics competition in 2018 as my project home automation select for the exhibition in Expocentre.
4.60+
23+ Reviews
62+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Add curved arrows to the following structures to show how electron pairs must be moved to interconvert the structures, and locate any formal charges. O-H -H
-
Which SN2 reaction of each pair would you expect to take place more rapidly in a protic solvent? (a) (1) Or (2) (b) (1) Or (2) (c) (1) Or (2) (d) (1) Or (2) Br (1.0M)-MeO(1.0M)-. / OMe + Br
-
The S N 2 reaction between a Grignard reagent and an epoxide works reasonably well when the epoxide is ethylene oxide. However, when the epoxide is substituted with groups that provide steric...
-
Marty owns an auto body repair shop. The business has no real value without him and the building is pretty much a mess after being full of paint fumes for all those years but the land is worth...
-
Briefly compare the advantages and disadvantages of illustrating data with charts (bar and line) versus tables.
-
The frame is used to support a 2-in.-thick plywood floor of a residential dwelling. Sketch the loading that acts along members BG and ABCD. Set a = 5 ft, b = 15ft. D E B G
-
I dont know you, and I very much liked working with Oscar Jones. Why was he transferred to Chicago? The customer you have just met for the first time is unhappy because of his relationship with the...
-
Outdoor Living has the following accounts receivable at year end, broken down by age: Prior experience has shown that the company will probably collect 95% of its current receivables. Furthermore,...
-
[50 pts] Write an assembly program that performs multiplication by addition: a. First, you should define a variable which is a string in the format of "a*b" where a and b represent numbers from 0 to...
-
Australian Flying School (AFS) is a company that provides aircraft pilot training and specialist instruction services to individuals (clients) which was incorporated in the State of Victoria in 1980....
-
Propose structures for compounds with the following 1H NMR spectra: (a) C 5 H 9 ClO 2 ? ?IR: 1735 cm ?1 ? (b) C 7 H 12 O 4 ? ? ?IR: 1735 cm ?1 ? (c) C 11 H 12 O 2 ? ??IR: 1710 cm ?1 ? TMS 8. 6. O ppm...
-
In the iodoform reaction, a triiodomethyl ketone reacts with aqueous NaOH to yield a carboxylate ion and iodoform (triiodomethane). Propose a mechanism for thisreaction. OH H20 Cl3 HCI3 R.
-
Fraudulent claims relative to the earned income tax credit appear to be significant in frequency, so the IRS audits at a higher rate the Forms 1040 that include the credit. This leaves the agency...
-
How to journal company recieved and paid septembers power bill?
-
Show how successfully its business performed during a period of time home depot reports its revenues and expenses?
-
Determining the effect of a transaction on the accounts is a desired outcome of which step of the recording process?
-
The following items are taken from the adjusted trial balance prepared as of December 3 1 , 2 0 1 7 . All accounts have normal balances. List of items taken from the adjusted trial balance. Cash $ 4...
-
6. Two of the three swimming pools in Keiko's town are owned and managed by the local town government. The town has decided to close both public pools permanently. Many people in the town are unhappy...
-
Use the data in Exercise 3.29 to make a scatter diagram with the P/E ratio on the horizontal axis and the return on the vertical axis. Does there appear to be a positive relationship, negative...
-
A condenser (heat exchanger) brings 1 kg/s water flow at 10 kPa quality 95% to saturated liquid at 10 kPa, as shown in Fig. P4.91. The cooling is done by lake water at 20C that returns to the lake at...
-
What is a chemical bond according to MO theory?
-
Using benzene and any other reagents, outline a synthesis of each of the following compounds. Tert-butylcyclohexane
-
Give the products expected (if any) when nitrobenzene reacts under the following conditions. (a) Cl2, FeCl3, heat (b) Fuming HNO3, H2SO4 (c) H3C-C-CI, AICI,( 1.1 equiv.), then H2O
-
Explain how you would distinguish between ethylbenzene, p-xylene, and styrene solely by NMR spectroscopy.
-
Question: Part One: Memo Write an analysis of the impact of tax laws and incentives as demonstrated in this current years tax return. Specifically, you must address the following: Explain the...
-
From the following ledger balances, prepare a trial balance for the Cheyenne Corp. at June 3 0 , 2 0 2 2 . All account balances are normal.Accounts Payable $ 1 0 , 0 0 0 , Cash $ 7 , 4 0 0 , Common...
-
Kathy is mixing fruit punch in a 32-cup punch bowl for a party. She plans to add at least 20 cups of fruit juice to the bowl before adding ginger ale. Identify the graph that represents the amounts...

Study smarter with the SolutionInn App


