Estimate the maximum heat flux obtainable with nucleate pool boiling on a clean surface for(a) Water at
Question:
Estimate the maximum heat flux obtainable with nucleate pool boiling on a clean surface for(a) Water at 1 atm on brass,(b) Water at 10 atm on brass.GIVENNucleate pool boiling on a clean surfaceASSUMPTIONSWater is at saturationtemperature
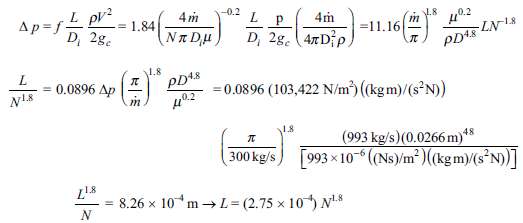
Transcribed Image Text:
L pV? D, 2g. -0.2 L.8 Ap=f = 1.84 =11.16 LN18 NR Du D, 2g. (47 Dip. PD48 1.8 PD48 =0.0896 (103,422 N/m) (kgm)/(s N)) 0.0896 Ap (993 kg/s)(0.0266 m)*s 300 kg/s) [993 x 10- (Ns)/m² )(kgm)/(s³N)] 48 8.26 x 10*m → L = (2.75 x 10) N*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
The maximum heat flux ...View the full answer

Answered By

Hillary Waliaulah
As a tutor, I am that experienced with over 5 years. With this, I am capable of handling a variety of subjects.
5.00+
17+ Reviews
30+ Question Solved
Related Book For 

Principles of heat transfer
ISBN: 978-0495667704
7th Edition
Authors: Frank Kreith, Raj M. Manglik, Mark S. Bohn
Question Posted:
Students also viewed these Mechanical Engineering questions
-
Estimate the nucleate-pool-boiling heat flux for water at 1 atm in contact with ground and polished stainless steel and Tx =17oC.
-
The solubility of Xe in water at 1 atm pressure and 20 oC is approximately 5 10-3. Compare this with the solubilities of Ar and Kr in water (Table 13.1) and explain what properties of the rare gas...
-
Water at 1 atm flows in a 1.25-cm-diameter brass tube at a velocity of 1.2 m/s. The tube wall is maintained at 110oC, and the average bulk temperature of the water is 96oC. Calculate the...
-
The management of a New York area investment firm wants to find out about the investment needs of its existing customers, for which it has an extensively detailed list, as a function of their...
-
On July 1, 2016, the Morgan County School District received a $50,000 gift from a local civic organization with the stipulation that, on June 30 of each year, $3,500 plus any interest earnings on the...
-
In problem, find the center and radius of each circle. Graph each circle. Find the intercepts, if any, of each circle. x 2 + (y - 1) 2 = 4
-
The 2014-T6 aluminum hollow column is fixed at its base and free at its top. Determine the maximum eccentric force \(P\) that can be safely supported by the column. Use the allowable stress method....
-
Freeman Electronics currently produces the shipping containers it uses to deliver the electronics products it sells. The monthly cost of producing 10,000 containers follows. Unit-level materials...
-
Entertainment technology introduction and conclusion? What is cloud gaming ? How has cloud gaming impacted the sector in terms of the H&T industry? How has tourism sector adapted to cloud gaming...
-
Complete Tsate's Form 1040-SR, Schedules A, B and D, Form 8949, Form 6252 and Qualified Dividends and Capital Gain Tax Worksheet. Tsate Kongia (birthdate 02/14/1954) is an unmarried high school...
-
Predict the nucleate-boiling heat transfer coefficient for water boiling at atmospheric pressure on the outside surface of a 1.5 cm OD vertical copper tube 1.5 m long. Assume the tube-surface...
-
Determine the excess temperature at one-half of the maximum heat flux for the fluid-surface combinations in Problem 10.3.From Problem 10.3: Estimate the maximum heat flux obtainable with nucleate...
-
Show that for motion in a straight line with constant acceleration a, initial velocity vo, and initial displacement so the displacement after time t is s = at2 + v0t + s0.
-
A child weighs 283 N. What is the child's mass in kilograms? kg What is the child's mass in slugs? slugs Need Help? Read It Submit Answer [-/2 Points] DETAILS Suppose an airline allows a maximum of...
-
When a business provides services for cash, what is the effect on the accounting equation are affected?
-
At Group One Company, when 4,000 units are produced: Fixed cost = $8 per unit Variable cost = $9 per unit. How much is the total cost if 2,700 units are produced? (Do not round intermediate...
-
Nora Incorporated sells a single product for $16. Variable costs include $6.08 for each unit plus a 6% sales commission. Fixed costs are $158,480 per month. Required: a. What is the contribution...
-
Gage Company reports the following information for its first year of operations: Units produced this year 7,000 units Units sold this year 6,500 units Direct materials $ 22 per unit Direct labor...
-
The \(\Lambda^{0}\) particle can be produced in pion-nucleon collisions according to the reaction \[\pi^{-}+p^{+} ightarrow \Lambda^{0}+K^{0}\] Show that the conservation laws for electric charge,...
-
A supermarket chain is interested in exploring the relationship between the sales of its store-brand canned vegetables (y), the amount spent on promotion of the vegetables in local newspapers (x1)...
-
Exactly 15 years from now, Jones will start receiving a pension of $20,000 a year. The payments will continue forever. How much is the pension worth now, assuming that the appropriate discount rate...
-
A long cylinder of 30-mm diameter, initia1ly at a uniform temperature of 1000 K. is suddenly quenched in a large, constant-temperature oil bath at 350 K. The cylinder properties are k = 1.7 W/m K, c...
-
A long pyroceram rod of diameter 20 mm is clad with a very thin metallic tube for mechanical protection. The bonding between the rod and the tube has a thermal contact resistance of R t.c = 0.12 m ...
-
A long rod 40 mm in diameter, fabricated from sapphire (aluminum oxide) and initially at a uniform temperature of 800 K, is suddenly cooled by a fluid at 300 K having a heat transfer coefficient of...
-
What is the difference between an intended strategy and a realized strategy? Provide examples of each type using Coca-Cola and Facebook.
-
Explain how improvements to organizational systems can ensure successful and sustained behavioral change. How would mitigate and remove any roadblocks in the change management process? What are plans...
-
Discuss and describe transactional leadership in term of management. ?

Study smarter with the SolutionInn App


