Experimental evidence indicates that the two oxygen of acetate ion are identical. Use resonance to explain this
Question:
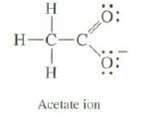
Experimental evidence indicates that the two oxygen of acetate ion are identical. Use resonance to explain this observation
Transcribed Image Text:
H H-C-C H Acetate ion Ö: O:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
The actual structure is a resonance hybrid o...View the full answer

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Experimental evidence indicates that most highly repetitive DNA sequences in the chromosomes of eukaryotes do not produce any RNA or protein products. What does this indicate about the function of...
-
What experimental evidence indicates that cyclin B is required for a cell to enter mitosis? What evidence indicates that cyclin B must be destroyed for a cell to exit mitosis?
-
A host of empirical evidence indicates that the gains from a typical merger accrue to the shareholders of the target corporation, not to the shareholders of the acquiring corporation. It seems the...
-
The cylinder in the figure has a radius of 2 ft and rolls without slipping on the horizontal surface. The bar AB is attached to the periphery of the cylinder by a pin at point A. The other end of the...
-
Swish Designs specialises in designing commercial office space in Auckland. The CEO, Ralph Polo has reviewed the financial results and has noticed that operating profits were below budget. He also...
-
Marvel Studio's motion picture Guardians of the Galaxy opened over the first two days of the 2014 Labor Day weekend to a record-breaking $94.3 million in ticket sales revenue in North America (the...
-
Jamie Walker, regional vice president of sales for American Food Processors (AFP), is looking at the performance numbers of his sales force for the past year. He is starting to get that sinking...
-
Water Technology, Inc. incurred the following costs during 20x1. The company sold all of its products manufactured during the year. Direct material...
-
Sodium chloride is the primary salt dissolved in ocean water. For every liter, there are about 35 grams of salt dissolved in ocean water, which corresponds to a salt concentration of 0.6 moles/liter,...
-
You want to determine the optimal tutor-to-student ratio. Students seeking tutoring will be randomly assigned to one of three groups: Group 1 will involve each tutor working with only one student; in...
-
Predict which of the following constitutional isomers for the compound that is formed from one atom each of hydrogen, oxygen, and chlorine is more stable: H-CI-: or H-O-CI:
-
Show the direction of the dipoles, if any, of these bonds: (a) C N (b) O N (c) O l (d) C Cl (e) B O (f) C Mg (g) C C (h) C H
-
In Problems 9 and 10, solve each equation in the complex number system. x 2 - 2x + 5 = 0
-
Model the system CO 2 (1) + tetralin(2) using k ij = 0.10. Generate a P-x-y diagram at 20C, 45C, and a T-x-y at 12 MPa and 22 MPa. What type of phase behavior does the system exhibit?
-
Most social scientists define the poor as being the lowest________ percent of our income recipients. a) 10 b) 20 c) 3 d) 40 e) 50
-
A force is described by the vector field \(\mathbf{F}=-\alpha\left(9 x^{2} \hat{\mathbf{i}}+2 z^{2} \hat{\mathbf{j}}+6 y z \hat{\mathbf{k}} ight)\). After obtaining the dimensions of \(\alpha\),...
-
The AF order parameter \(N_{z}\) is related to the coherent state order parameter \(\beta\) by \(\left\langle N_{z} ight angle=2 \Omega\left|b_{2} ight|\left(f-\beta^{2} ight)^{1 / 2} \beta\), where...
-
Suppose, in TCPs adaptive retransmission mechanism, that EstimatedRTT is 4.0 at some point and subsequent measured RTTs all are 1.0. How long does it take before the TimeOut value, as calculated by...
-
Alameda Ltd has two divisions, Division Alpha and Division Beta. Division Alpha makes widgets, which it can sell externally for $\$ 45$ as well as internally to Division Beta. The marginal cost of...
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
Find the solution of the equation cos x = x correct to two decimal places.
-
Draw a line-bond structure for vinyl chloride, C2H3C1, the starting material from which PVC [poly (vinyl chloride)] plastic is made.
-
Fill in any nonbonding valence electrons that are missing from the followingstructures: CH (b) (c) (a) " NH2 - Dimethyl disulfide Acetate ion Acetamide
-
Convert the following line-bond structures into molecularformulas: CH CH- (a) (b) . Hi H. c=C Vitamin C Aspirin (acetylsalicylic acid) (ascorbic acid) (c) H. - (d) H~ CH c-o H. . HOI I H H OH...
-
Shahid Pakistan Limited (SPL) is engaged in the production of three products: J, K and L. Following is the extract from its latest annual management accounts: Description J Products K Total L Units...
-
John Company produces hats and sells them for $100 each. His cost to produce the hats are: DM 20 per unit DL 30 per unit VMOH 10 per unit FMOH 40,000 Selling expenses are $5 per unit and are all...
-
John Company produces hats and sells them for $100 each. His cost to produce the hats are: DM 20 per unit DL 30 per unit VMOH 10 per unit FMOH 40,000 Selling expenses are $5 per unit and are all...

Study smarter with the SolutionInn App


